Chapter 9: Q13. (page 347)
Question: Draw the structure of each transition state in the two-step mechanism for the reaction.
CH3CH2OH + H2So4 → CH3CH=CH2+ H2O
Short Answer
Answer
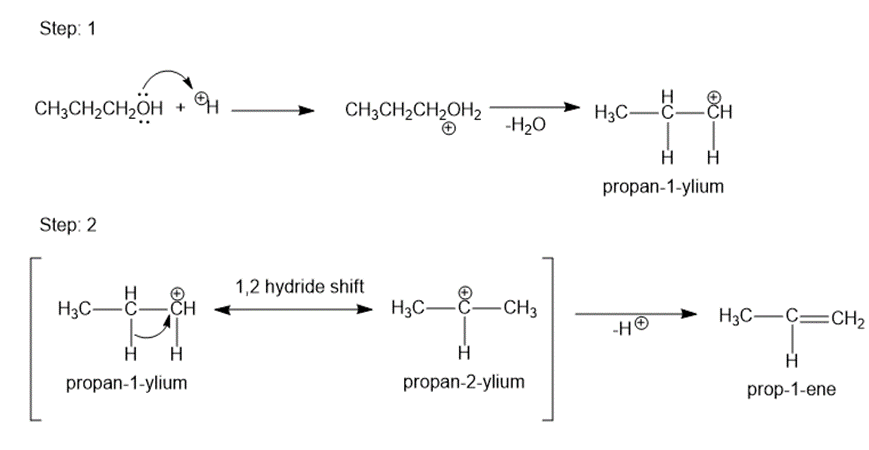
The mechanism is shown below.
Learning Materials
Features
Discover
Chapter 9: Q13. (page 347)
Question: Draw the structure of each transition state in the two-step mechanism for the reaction.
CH3CH2OH + H2So4 → CH3CH=CH2+ H2O
Answer
The mechanism is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Prepare each compound from cyclopentanol. More than one step may be needed.
a.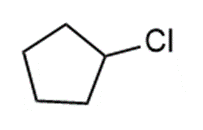
b. 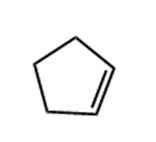
c. 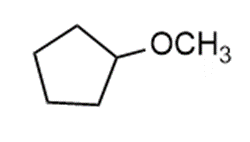
d. 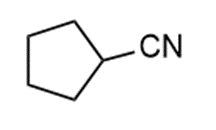
Question: Explain the following observation. When 3-methylbutan-2-ol is treated with HBr, a single alkylbromide is isolated, resulting from a 1,2-shift. When 2-methylpropan-1-ol is treated with HBr, no rearrangement occurs to form an alkyl bromide.
Question: Answer each question using the ball-and-stick model of compound A.
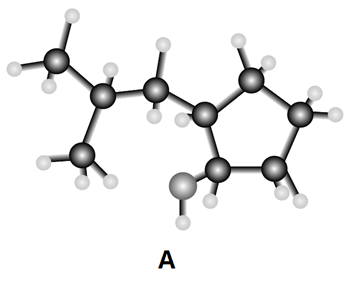
a. Give the IUPAC name for A, including R,Sdesignations for stereogenic centers.
b. Classify A as a alcohol.
c. Draw a stereoisomer for A and give its IUPAC name.
d. Draw a constitutional isomer that contains an OH group and give its IUPAC name.
e. Draw a constitutional isomer that contains an ether and give its IUPAC name.
f. Draw the products formed (including stereochemistry) when A is
treated with each reagent: [1] NaH; [2] ; [3] , pyridine;
[4] HCl; [5] ,pyridine; [6] TsCl, pyridine.
Question: Show how a 1,2-shift forms a more stable carbocation from each intermediate.
a.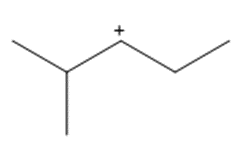
b. 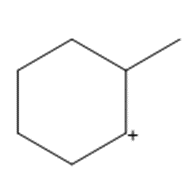
c. 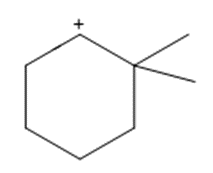
Question: Draw the products of each reaction, indicating the stereochemistry around any stereogenic centers.
a. 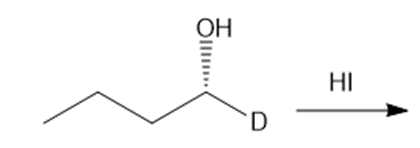
b. 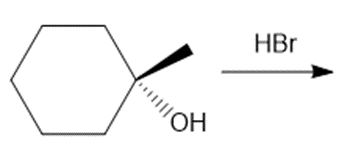
c.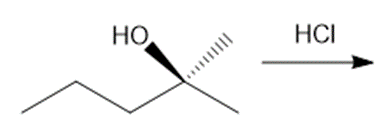
What do you think about this solution?
We value your feedback to improve our textbook solutions.