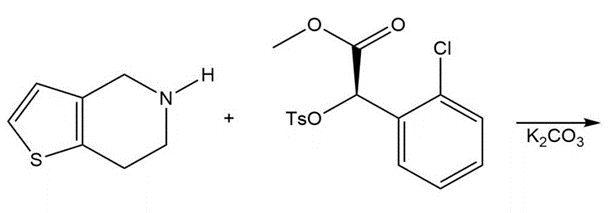
Chapter 9: Q49. (page 377)
Question: Draw the product of the following reaction, one step in the synthesis of the antiplatelet agent clopidogrel used to reduce the risk of stokes.
Short Answer
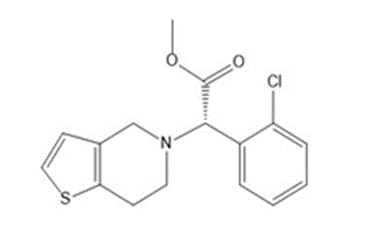
Answer
Learning Materials
Features
Discover
Chapter 9: Q49. (page 377)
Question: Draw the product of the following reaction, one step in the synthesis of the antiplatelet agent clopidogrel used to reduce the risk of stokes.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Give the IUPAC name for each alcohol.
a. 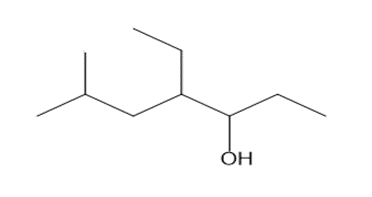
b. 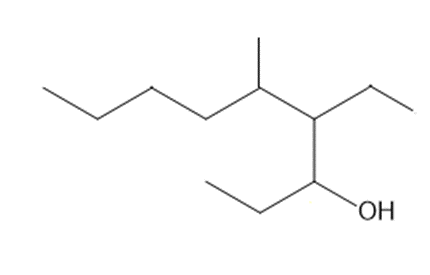
c. 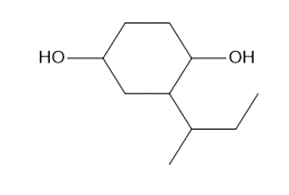
d. 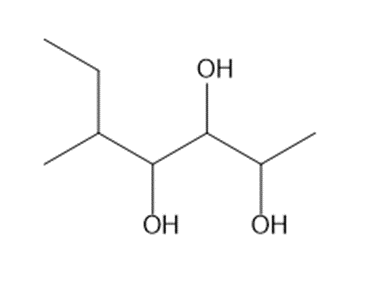
e.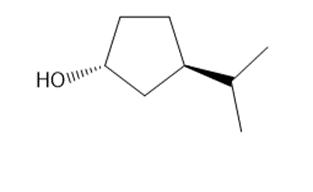
Question:Rearrangements can occur during the dehydration of alcohols even though no carbocation is formed-that is, a 1,2-shift occurs as the bond is broken, forming a more stable carbocation, as shown. Using this information, draw a stepwise mechanism that shows how is dehydrated with to form a mixture of and the cis and trans isomers of . We will see another example of this type of rearrangement in Section 18.5C.
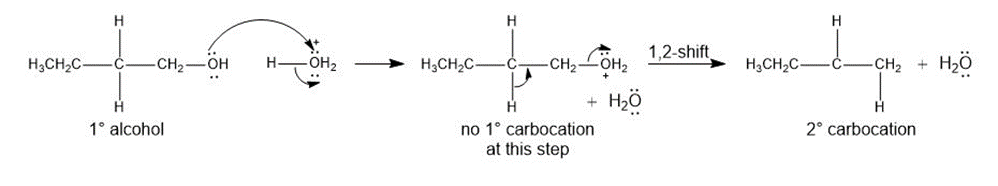
Question: Draw the organic product(s) formed when is treated with each reagent.
a.
b. NaH
c. HCl +
d. HBr
e., pyridine
f.
g. TsCl, pyridine
h. [1] NaH; [2]
i. [1]TsCl, pyridine;[2] NaSH
j. , pyridine
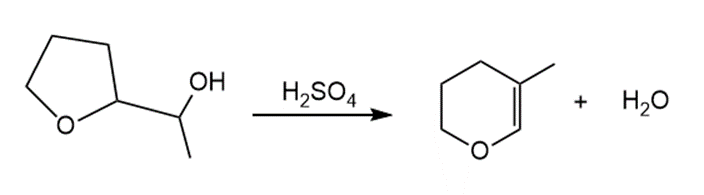
Question: Draw a stepwise, detailed mechanism for the following reaction.
Question: Draw two steps to convert (CH3)3CHOH into each of the following compounds: (CH3)2N3 , and (CH3)2CHOCH2CH3.
What do you think about this solution?
We value your feedback to improve our textbook solutions.