Chapter 9: Q48. (page 337)
Question: Draw the products of each reaction and indicate stereochemistry around stereogeniccenters.
a.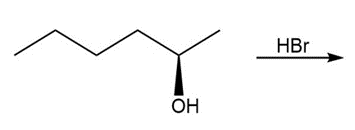
b. 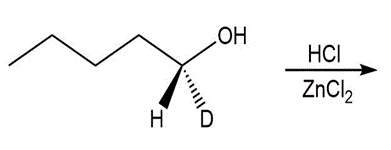
c. 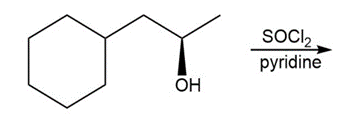
d. 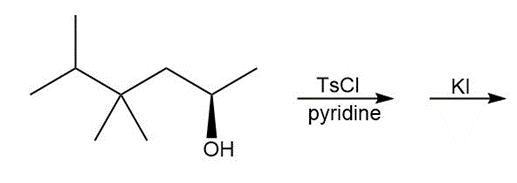
Short Answer
Answer
a. 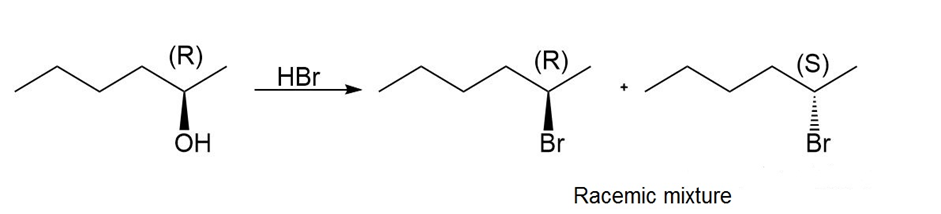
b
c. 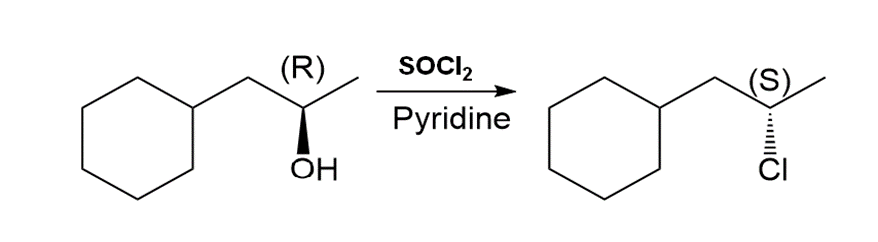
d.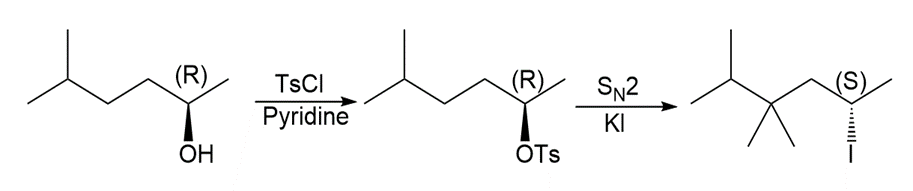
Learning Materials
Features
Discover
Chapter 9: Q48. (page 337)
Question: Draw the products of each reaction and indicate stereochemistry around stereogeniccenters.
a.
b. 
c. 
d. 
Answer
a. 
b
c. 
d.
All the tools & learning materials you need for study success - in one app.
Get started for free
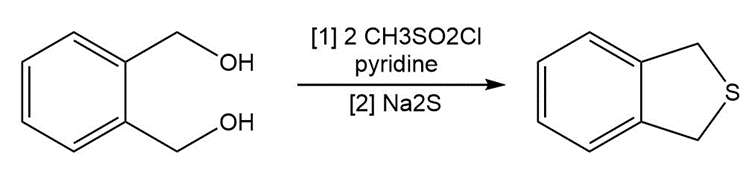
Question: The following two-step procedure was used to prepare a sulfide from a diol. Draw theintermediate formed in Reaction [1] and draw a mechanism for Reaction [2].
Question: If the reaction of an alcohol with SOCl2and pyridine follows an SN2 mechanism, what is the stereochemistry of the alkyl chloride formed from (R)-butan-2-ol?
Question: Draw the products of each reaction, indicating the stereochemistry around any stereogenic centers.
a. 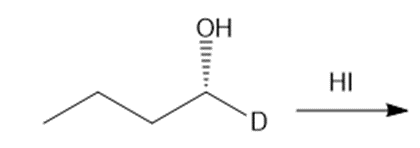
b. 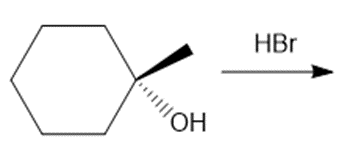
c.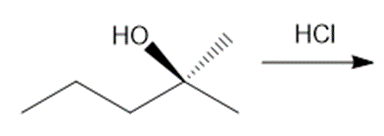
Question: Give the structure corresponding to each name.
a. 7,7-dimethyloctan-4-ol
b. 5-methyl-4-propylheptan-3-ol
c. 2-tert-butyl-3-methylcyclohexanol
d. trans-cyclohexane-1,2-diol
Question: If the reaction of an alcohol with follows an mechanism, what is the stereochemistry of the alkyl bromide formed from (R)-butan-2-ol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.