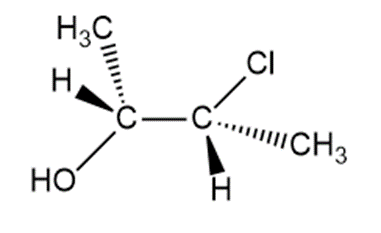
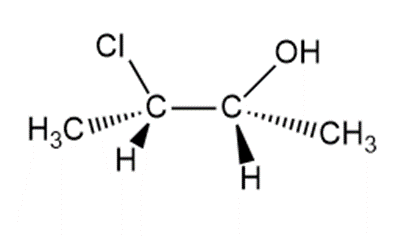
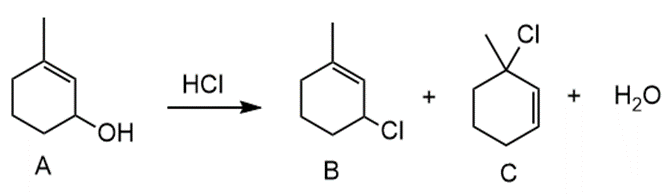
Chapter 9: Q70. (page 380)
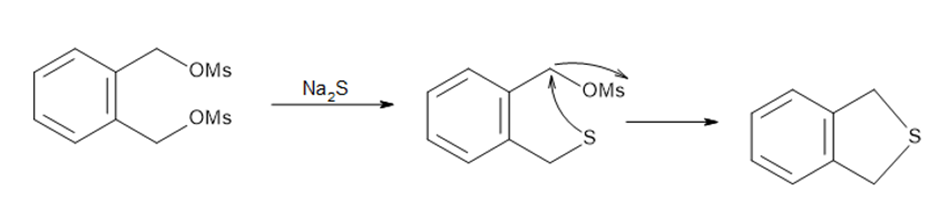
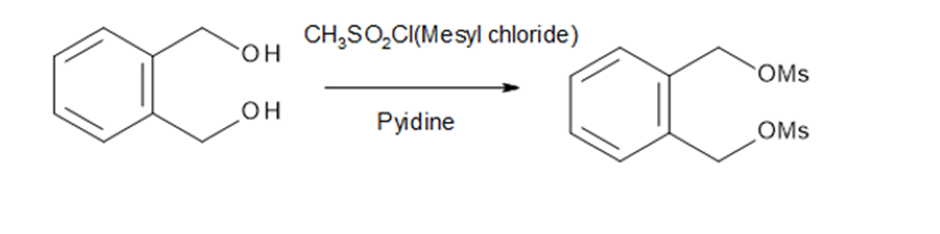
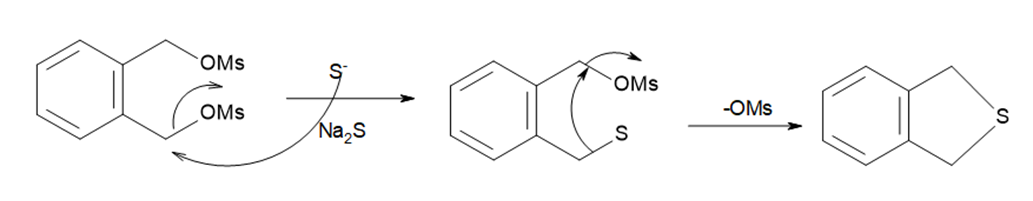
Question: The following two-step procedure was used to prepare a sulfide from a diol. Draw theintermediate formed in Reaction [1] and draw a mechanism for Reaction [2].
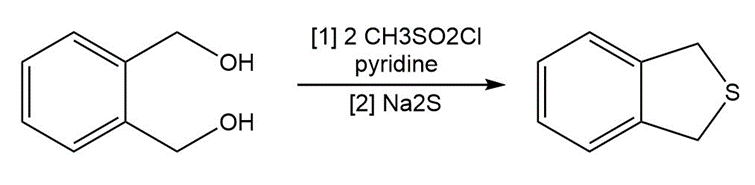
Short Answer
Answer
The intermediate formed in reaction [1]
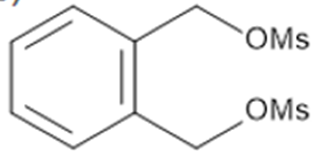
The mechanism of reaction [2]