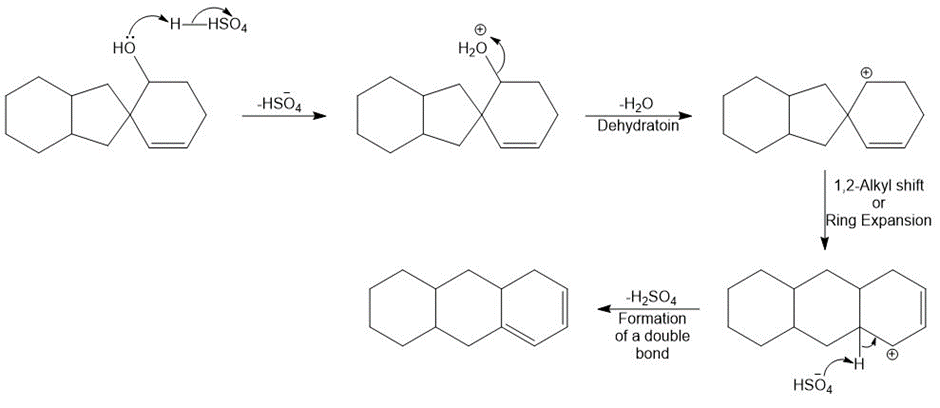
Chapter 9: Q77. (page 381)
Question: Draw a stepwise, detailed mechanism for the following reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 9: Q77. (page 381)
Question: Draw a stepwise, detailed mechanism for the following reaction.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
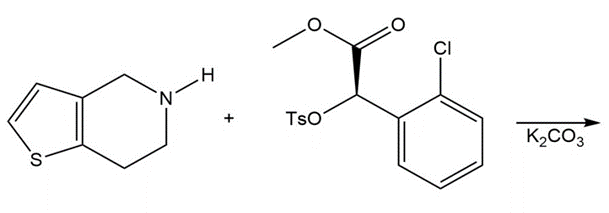
Question: Draw the product of the following reaction, one step in the synthesis of the antiplatelet agent clopidogrel used to reduce the risk of stokes.
Question: Draw the products of each reaction and indicate stereochemistry around stereogeniccenters.
a.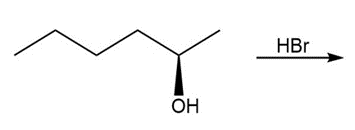
b. 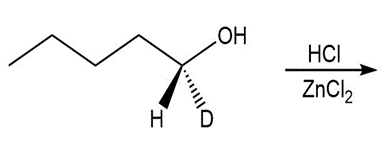
c. 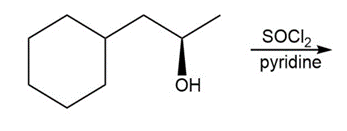
d. 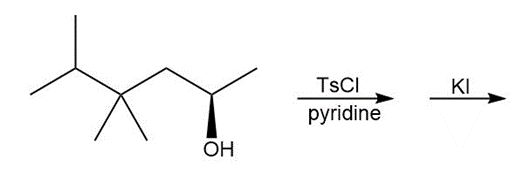
Question: What alkenes are formed when each alcohol is dehydrated with TsOH? Label the major product when a mixture results.
a. 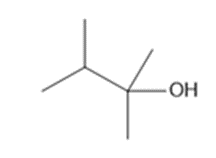
b. 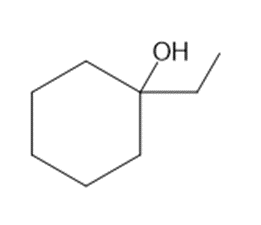
c. 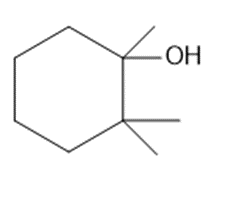
d. 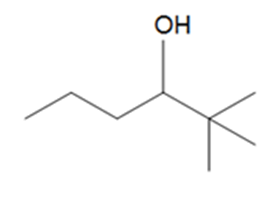
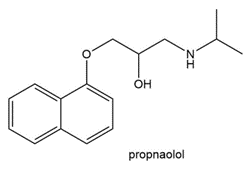
Question:Propranolol, an antihypertensive agent used in the treatment of high blood pressure, canbe prepared from 1-naphthol, epichlorohydrin, and isopropyl amine using two successivenucleophilic substitution reactions. Devise a stepwise synthesis of propranolol from thesestarting materials.
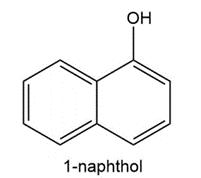
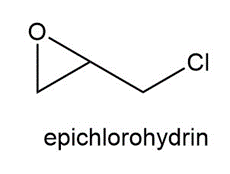
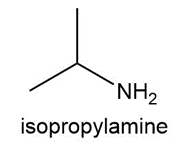
Question: Explain why two substitution products are formed in the following reaction.
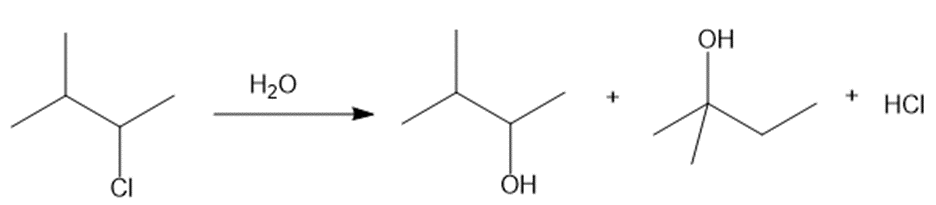
What do you think about this solution?
We value your feedback to improve our textbook solutions.