Chapter 9: Q47. (page 377)
Question: Draw the products formed when is treated with each reagent.
Short Answer
Answer
a. 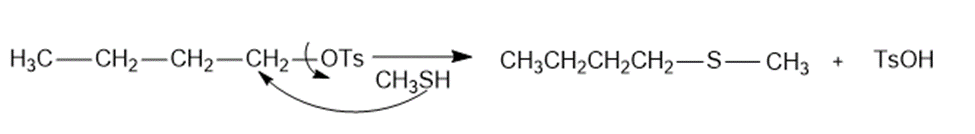
b. 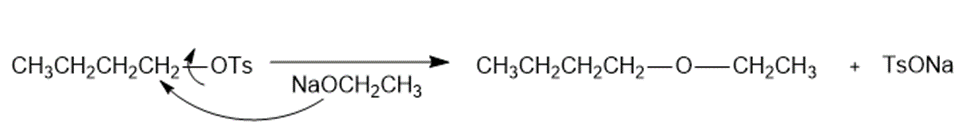
c. 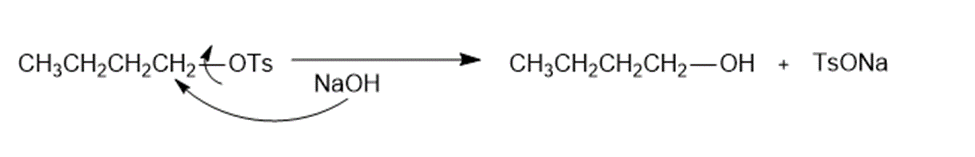
d.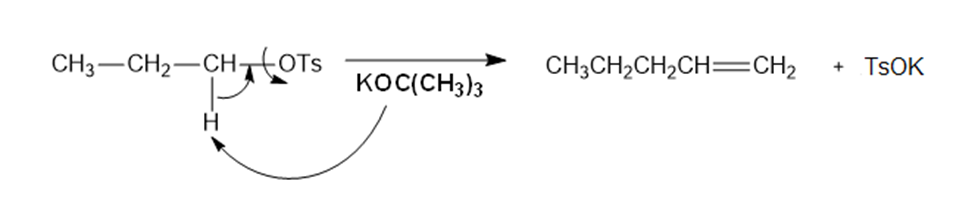
Learning Materials
Features
Discover
Chapter 9: Q47. (page 377)
Question: Draw the products formed when is treated with each reagent.
Answer
a. 
b. 
c. 
d.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which mechanism is favored by the use of crown ethers in nonpolar solvents, ?
Question: Draw the products of each reaction.
a. 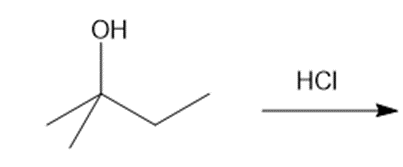
b. 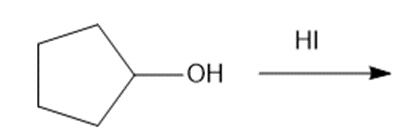
c.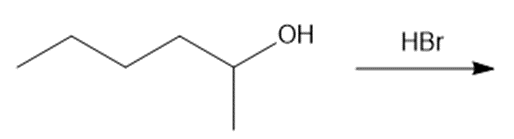
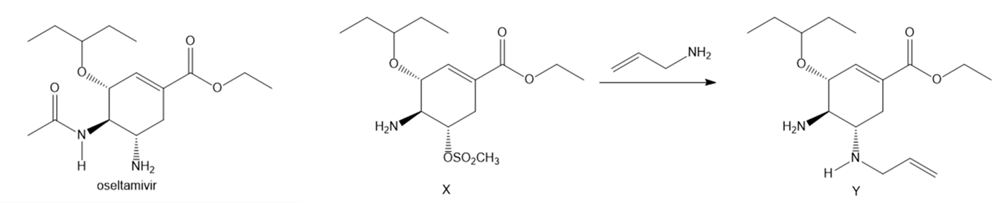
Question: Aziridines are heterocycles that contain an N atom in a three-membered ring. Like epoxides, aziridines are strained and reactive because the 60° bond angles of the three-membered ring deviate greatly from the theoretical tetrahedral bond angle. One step in the synthesis of the drug oseltamivir (trade name Tamiflu, Section 3.2) involves the conversion of amine X to diamine Y, a reaction that occurs by way of an intermediate aziridine. Draw a stepwise mechanism for the conversion of X to Y. Indicate the structure of the aziridine intermediate, and explain the trans stereochemistry of the two amines in Y
Question: Explain why it is not possible to prepare tert-butyl phenyl ether using a Williamson ether synthesis.
Question: Draw the products of each reaction and indicate the stereochemistry where appropriate.
a. 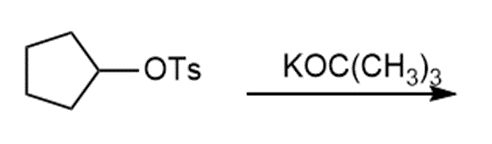
b. 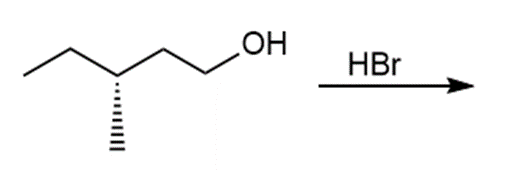
c. 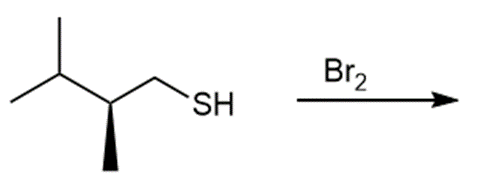
d. 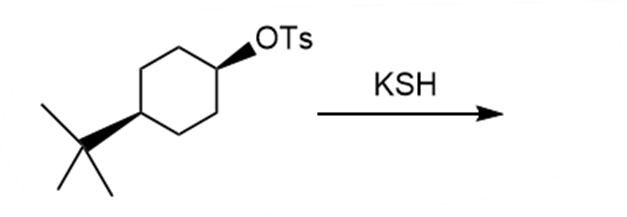
e. 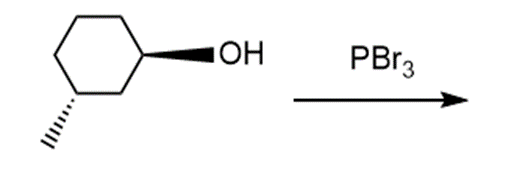
f. 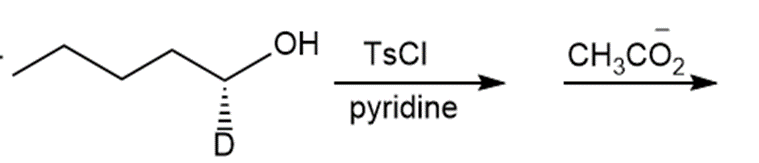
g. 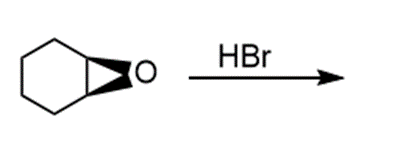
h. 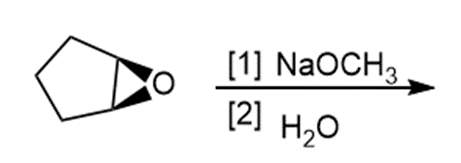
i. 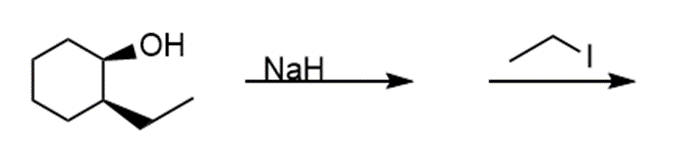
j. 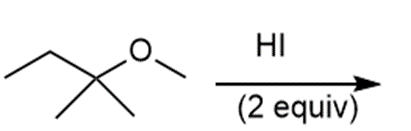
k. 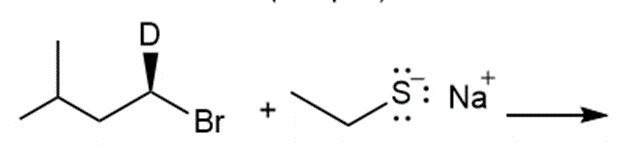
l. 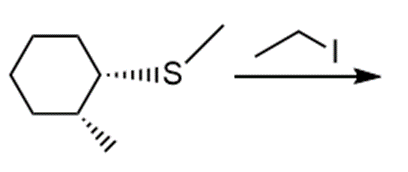
What do you think about this solution?
We value your feedback to improve our textbook solutions.