Chapter 9: Q57. (page 378)
Question: Draw a stepwise, detailed mechanism for the following reaction.
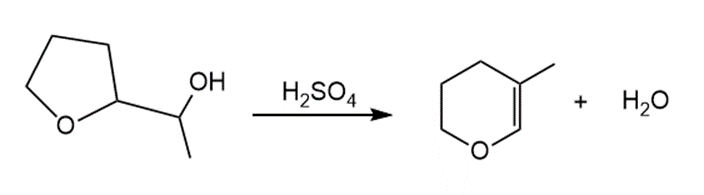
Short Answer
Answer
The mechanism of the reaction is as shown below.
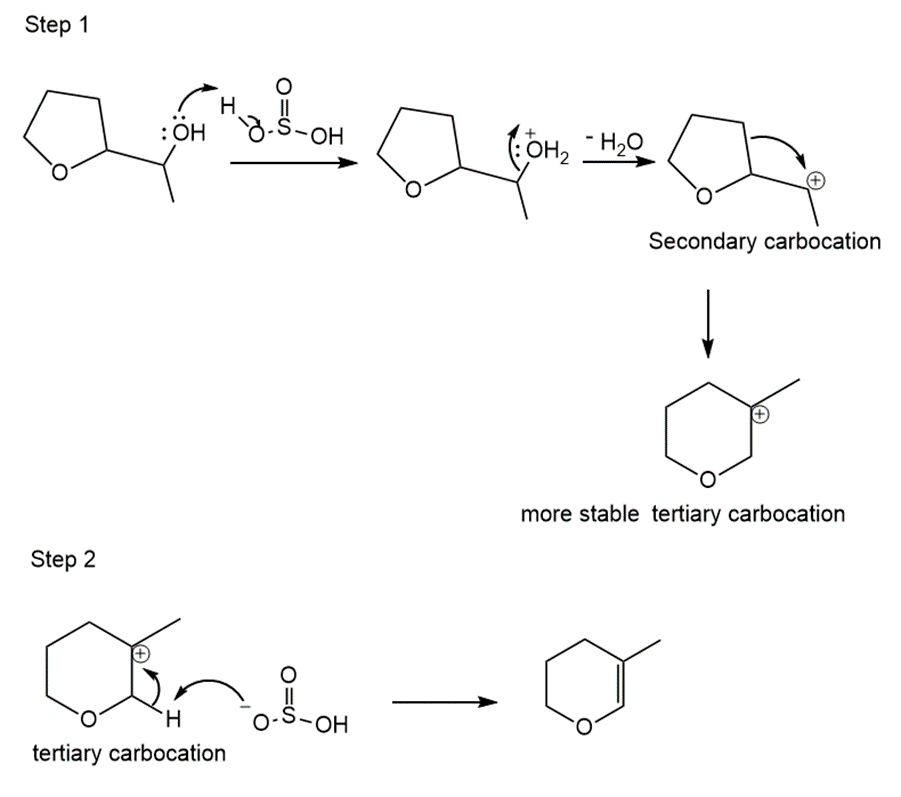
Learning Materials
Features
Discover
Chapter 9: Q57. (page 378)
Question: Draw a stepwise, detailed mechanism for the following reaction.

Answer
The mechanism of the reaction is as shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What alkenes are formed when each alcohol is dehydrated with TsOH? Label the major product when a mixture results.
a. 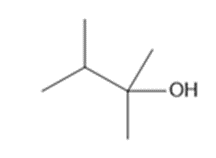
b. 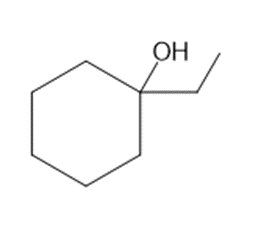
c. 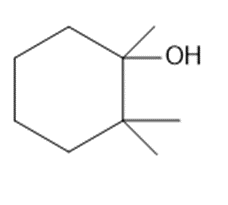
d. 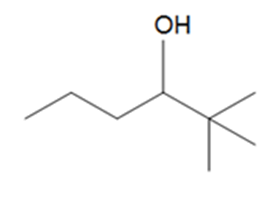
Question: Which mechanism is favored by the use of crown ethers in nonpolar solvents, ?
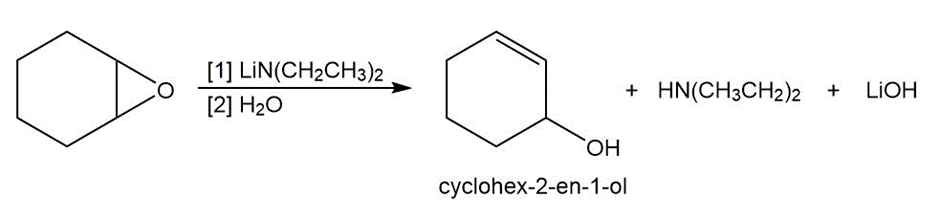
Question: Epoxides are converted to allylic alcohols with non-nucleophilic bases such as lithiumdiethylamide . Draw a stepwise mechanism for the conversion of1,2-epoxycyclohexane to cyclohex-2-en-1-ol with this base. Explain why a strong bulky basemust be used in this reaction.
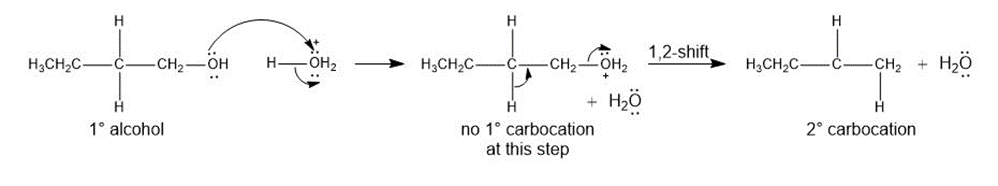
Question:Rearrangements can occur during the dehydration of alcohols even though no carbocation is formed-that is, a 1,2-shift occurs as the bond is broken, forming a more stable carbocation, as shown. Using this information, draw a stepwise mechanism that shows how is dehydrated with to form a mixture of and the cis and trans isomers of . We will see another example of this type of rearrangement in Section 18.5C.
Question: Why is the boiling point of propane-1,3-diol higher than the boiling point of propane-1,2-diol role="math" localid="1648635549686" ? Why do both diols have a higher boiling point than butan-1-ol ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.