Chapter 9: Q1. (page 333)
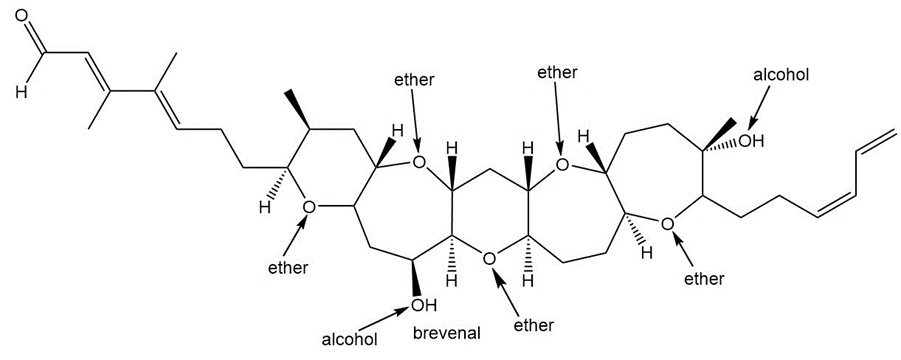
Question: Label each ether and alcohol in Brevenal, a marine natural product. Classify each alcohol as
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 9: Q1. (page 333)
Question: Label each ether and alcohol in Brevenal, a marine natural product. Classify each alcohol as

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: 1,2-Diols are converted to carbonyl compounds when treated with strong acids, in a reaction called the pinacol rearrangement.
(a) Draw a stepwise mechanism for this reaction. (Hint: The reaction proceeds by way of carbocation intermediates.)
(b) Assuming that the pinacol rearrangement occurs via the more stable carbocation, draw the rearrangement product formed from diol D.
Question: a) What is the major alkene formed when A is dehydrated with ? (b) What is the major alkene formed when A is treated with and pyridine? Explain why the major product is different in these reactions.
Question: Draw the products of each reaction, indicating the stereochemistry around any stereogenic centers.
a. 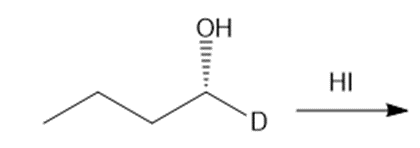
b. 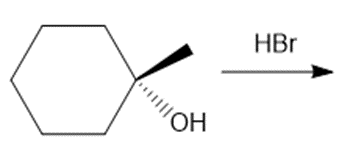
c.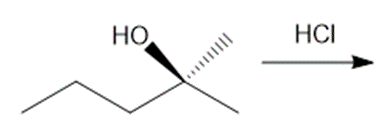
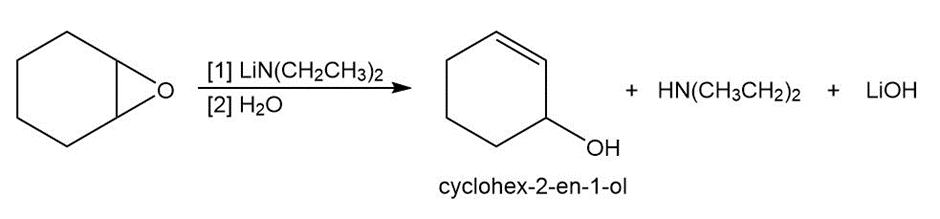
Question: Epoxides are converted to allylic alcohols with non-nucleophilic bases such as lithiumdiethylamide . Draw a stepwise mechanism for the conversion of1,2-epoxycyclohexane to cyclohex-2-en-1-ol with this base. Explain why a strong bulky basemust be used in this reaction.
Question: Draw two steps to convert (CH3)3CHOH into each of the following compounds: (CH3)2N3 , and (CH3)2CHOCH2CH3.
What do you think about this solution?
We value your feedback to improve our textbook solutions.