Chapter 9: Q80. (page 382)
Question: Draw a stepwise mechanism for the following reaction.
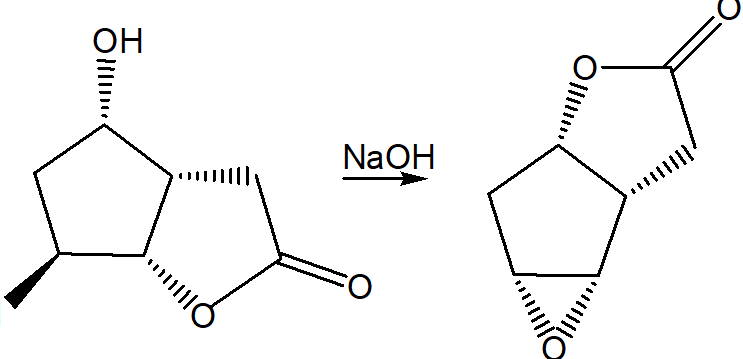
Short Answer
Answer
The stepwise mechanism of the given reaction is shown hereunder.
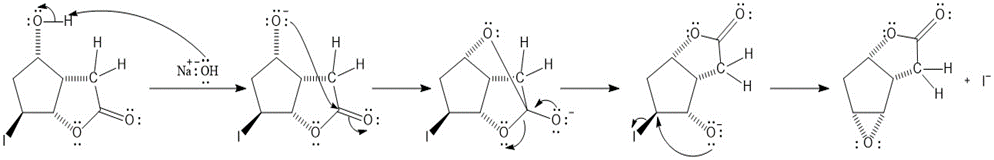
Mechanism
Learning Materials
Features
Discover
Chapter 9: Q80. (page 382)
Question: Draw a stepwise mechanism for the following reaction.

Answer
The stepwise mechanism of the given reaction is shown hereunder.

Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the organic product(s) formed when is treated with each reagent.
a.
b. NaH
c. HCl +
d. HBr
e., pyridine
f.
g. TsCl, pyridine
h. [1] NaH; [2]
i. [1]TsCl, pyridine;[2] NaSH
j. , pyridine
Question: Draw the products of each reaction and indicate the stereochemistry where appropriate.
a. 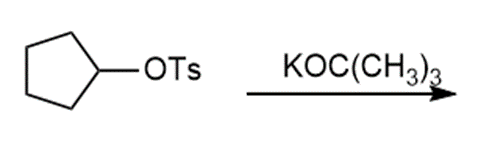
b. 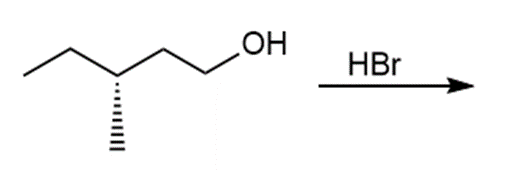
c. 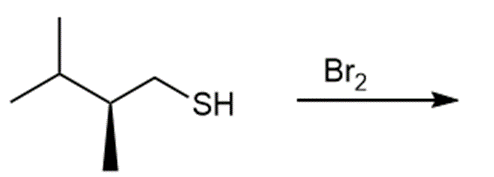
d. 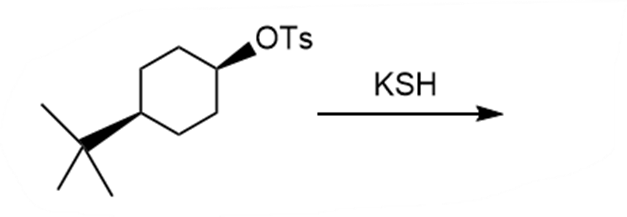
e. 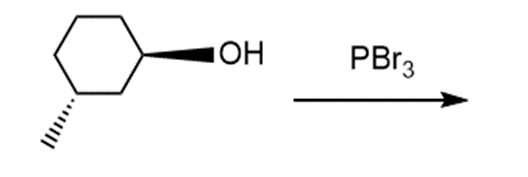
f. 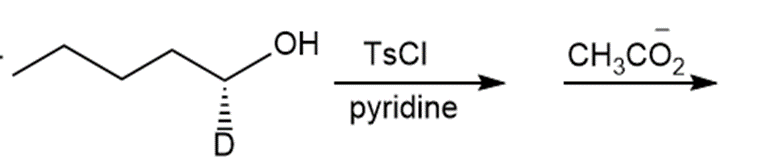
g. 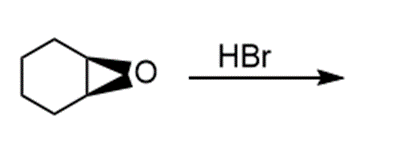
h. 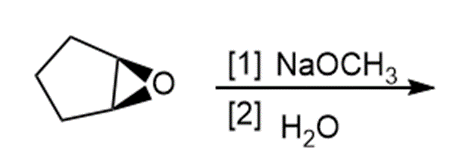
i. 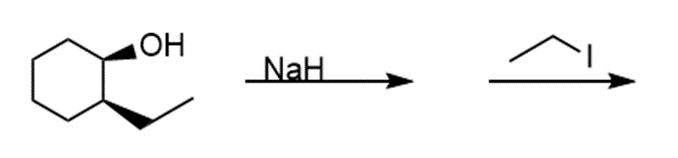
j. 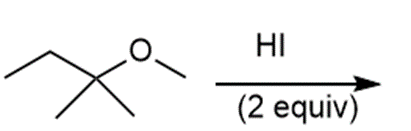
k. 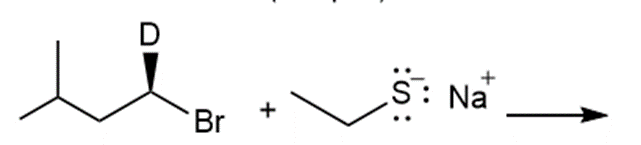
l. 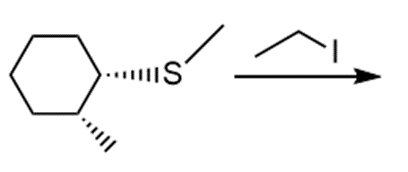
Question: Draw a stepwise mechanism for each reaction
a. 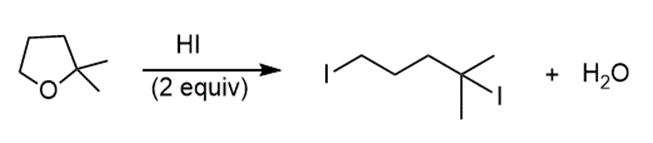
b. 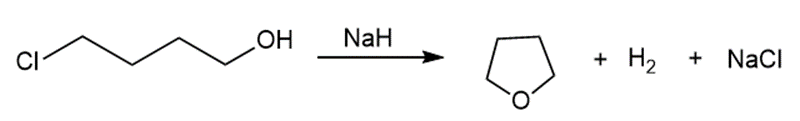
Question: If the reaction of an alcohol with follows an mechanism, what is the stereochemistry of the alkyl bromide formed from (R)-butan-2-ol?
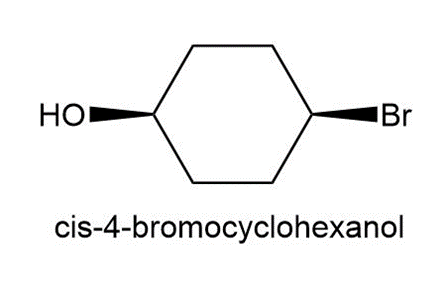
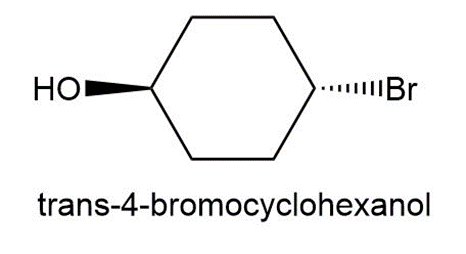
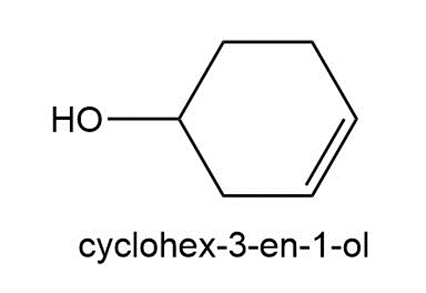
Question: Treatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4-bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation
What do you think about this solution?
We value your feedback to improve our textbook solutions.