Chapter 9: Q54. (page 378)
Question: Sometimes carbocation rearrangements can change the size of a ring. Draw a stepwise, detailed mechanism for the following reaction.
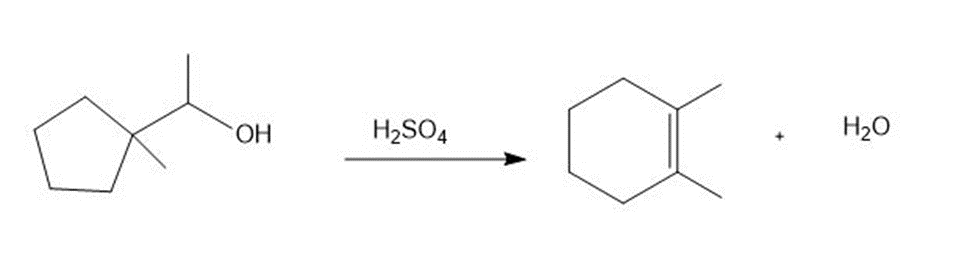
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 9: Q54. (page 378)
Question: Sometimes carbocation rearrangements can change the size of a ring. Draw a stepwise, detailed mechanism for the following reaction.

Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the products of each reaction and indicate the stereochemistry where appropriate.
a. 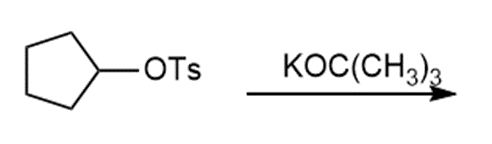
b. 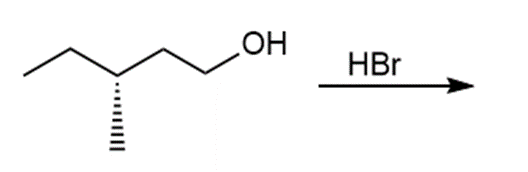
c. 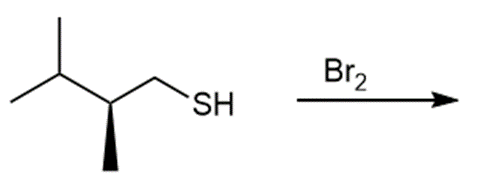
d. 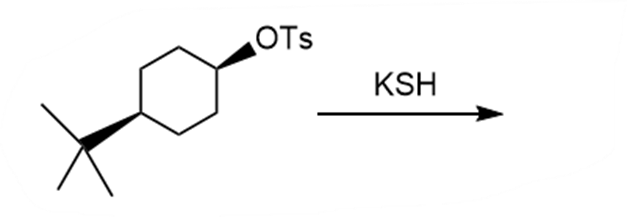
e. 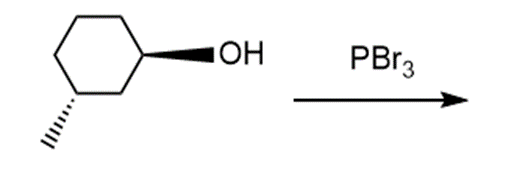
f. 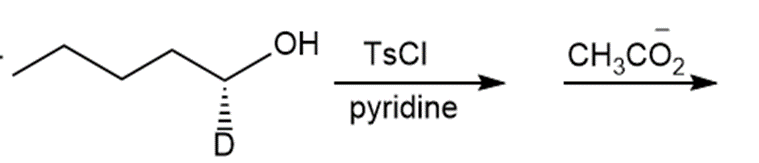
g. 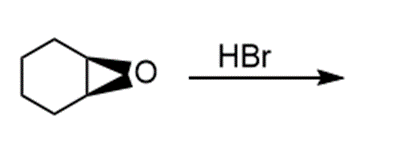
h. 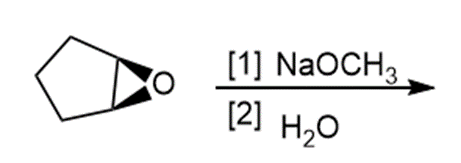
i. 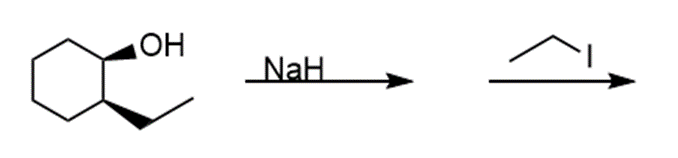
j. 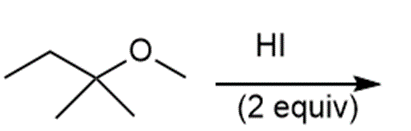
k. 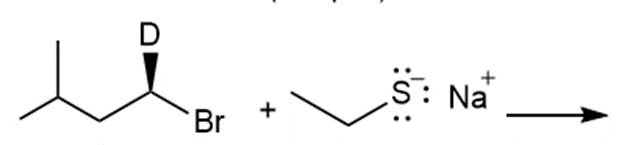
l. 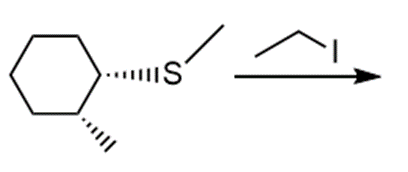
Question: Draw a stepwise mechanism for the following reaction.
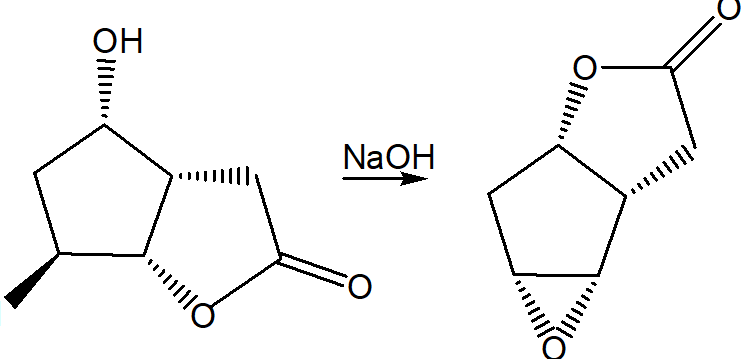
Question: Draw the substitution product formed (including stereochemistry) when (R)-hexan-2-ol is treated with each series of reagents: (a) NaH, followed by ; (b) TsCl and pyridine, followed by ; (c) , followed by . Which two routes produce identical products?
Question: Draw two different routes to each of the following ethers using a Williamson ether synthesis. Indicate the preferred route (if there is one).
a. 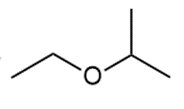
b. 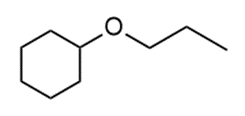
c. 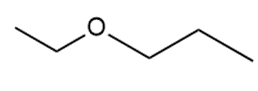
Question: Explain why it is not possible to prepare tert-butyl phenyl ether using a Williamson ether synthesis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.