Chapter 11: Q13 P. (page 333)
Predict whether each of the following substitution reactions is likely to be or:
(a)
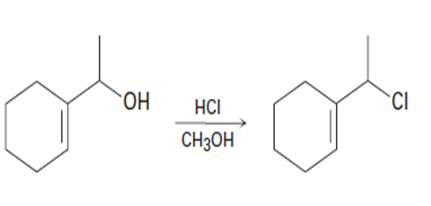
(b)
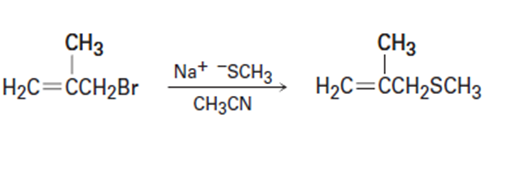
Short Answer
(a) This reaction occurs by an mechanism.
(b) This reaction occurs by an mechanism.
Learning Materials
Features
Discover
Chapter 11: Q13 P. (page 333)
Predict whether each of the following substitution reactions is likely to be or:
(a)

(b)

(a) This reaction occurs by an mechanism.
(b) This reaction occurs by an mechanism.
All the tools & learning materials you need for study success - in one app.
Get started for free
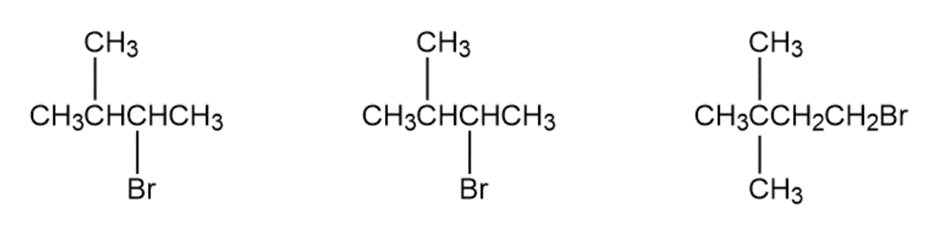
Question: Order each of the following sets of compounds with respect to reactivity:
b.
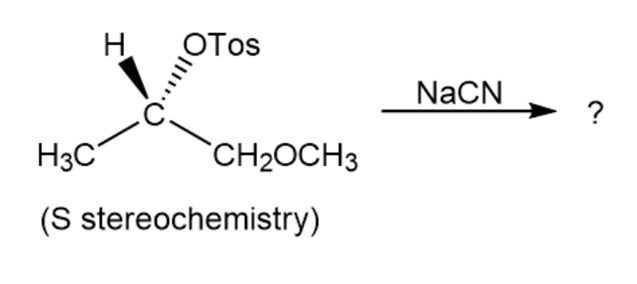
Reaction of the following S to sylate with cyanide ion yields a nitrileproduct that also has S stereochemistry. Explain.
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereochemistry of both reactant and product
Which reactant in each of the following pairs is more nucleophilic? Explain.
F)
Which reaction in each of the following pairs would you expect to be faster?
(b) Thedisplacement byon bromoethane or on bromocyclohexane
What do you think about this solution?
We value your feedback to improve our textbook solutions.