Chapter 11: Q59E (page 350)
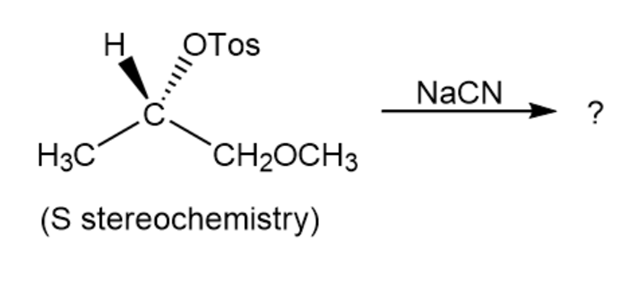
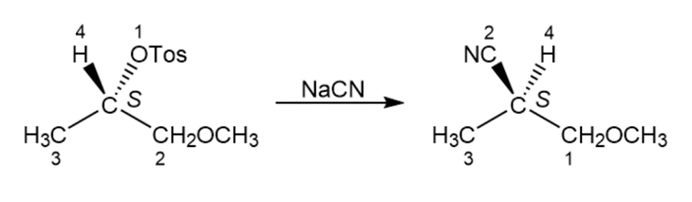
Reaction of the following S to sylate with cyanide ion yields a nitrileproduct that also has S stereochemistry. Explain.
Short Answer
Learning Materials
Features
Discover
Chapter 11: Q59E (page 350)
Reaction of the following S to sylate with cyanide ion yields a nitrileproduct that also has S stereochemistry. Explain.


All the tools & learning materials you need for study success - in one app.
Get started for free
Predict whether each of the following substitution reactions is likely to be or:
(a)
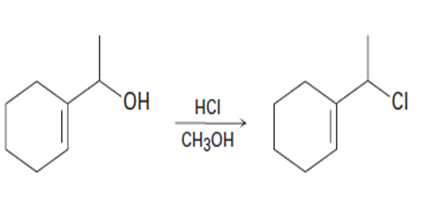
(b)
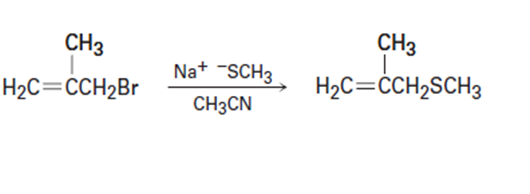
d) Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
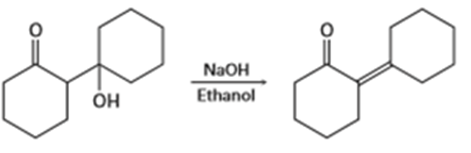
What effect would you expect the following changes to have on the rate of the reaction of ethanol with 2-iodo-2-methyl butane?
(b) The concentration of the ethanol is halved by adding diethyl ether as an inert solvent.
Rank the following substances in order of their expected reactivity:
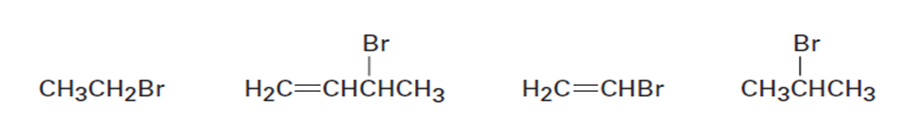
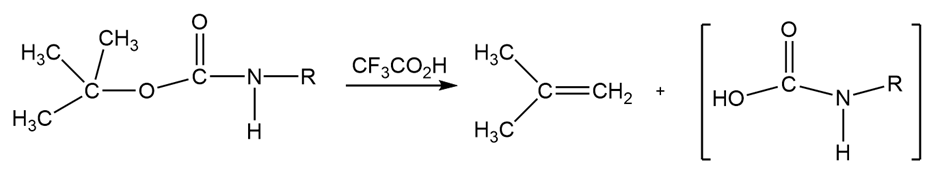
Propose a mechanism for the following reaction, an important step in the laboratory synthesis of proteins:
What do you think about this solution?
We value your feedback to improve our textbook solutions.