Chapter 11: 46bE (page 350)
Which reaction in each of the following pairs would you expect to be faster?
(b) Thedisplacement byon bromoethane or on bromocyclohexane
Short Answer
Bromoethanebromocyclohexane
Learning Materials
Features
Discover
Chapter 11: 46bE (page 350)
Which reaction in each of the following pairs would you expect to be faster?
(b) Thedisplacement byon bromoethane or on bromocyclohexane
Bromoethanebromocyclohexane
All the tools & learning materials you need for study success - in one app.
Get started for free
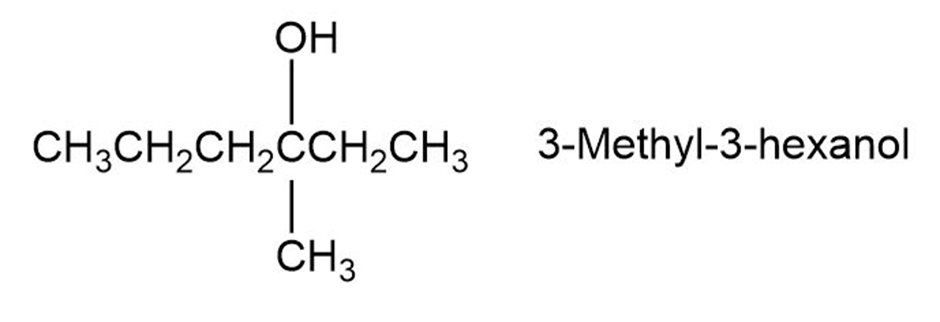
The reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-Bromo-3-methyl hexane. Explain.
Question: Propose structures for compounds that fit the following descriptions:
(a) An alkyl halide that gives a mixture of three alkenes on E2 reaction
Draw all isomers of ,name them, and arrange them in order of decreasing reactivity in thereaction:
Write the product you would expect from reaction of each of the following alkyl halides with (1)Na+-SCH3and (2)Na+-OH(green = Cl):
a)
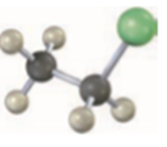
Question: Order each of the following sets of compounds with respect to reactivity:
c. CH3CH2 CH2OCH3 CH3CH2 CH2OTs CH3CH2 CH2Br
What do you think about this solution?
We value your feedback to improve our textbook solutions.