Chapter 11: Q11-71E (page 350)
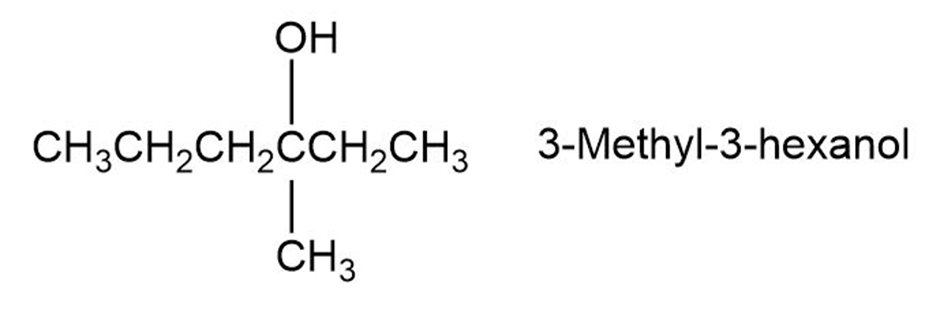
The reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-Bromo-3-methyl hexane. Explain.
Short Answer
 Formation of carbocation
Formation of carbocation
 Formation of the desired product
Formation of the desired product
Learning Materials
Features
Discover
Chapter 11: Q11-71E (page 350)
The reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-Bromo-3-methyl hexane. Explain.

 Formation of carbocation
Formation of carbocation
 Formation of the desired product
Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
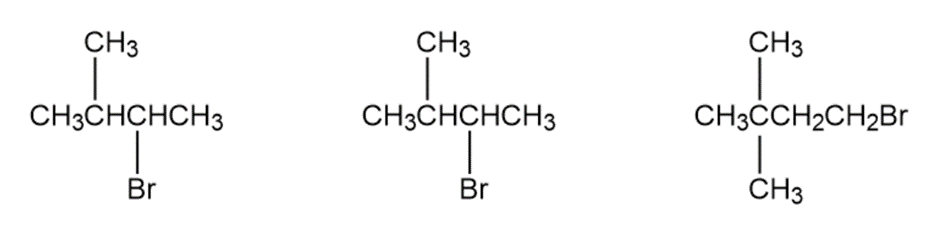
Question: Order each of the following sets of compounds with respect to reactivity:
b.
Question: (R)-2-Bromooctane undergoes racemization to give ()-2-bromooctane when treated with NaBr in dimethyl sulfoxide. Explain.
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereochemistry of both reactant and product
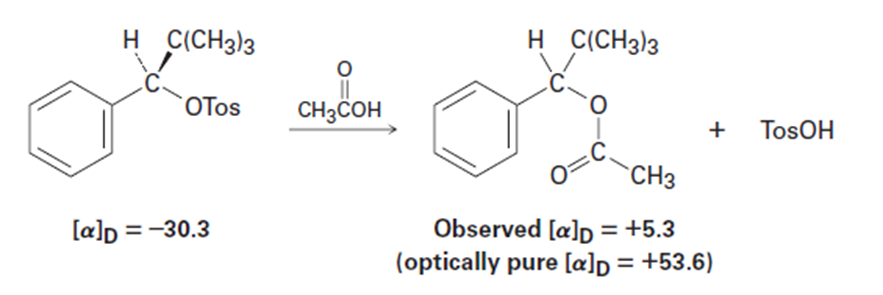
Among the many examples of reactions that occur with incomplete racemization, the optically pure tosylate of 2,2-dimethyl-1-phenyl-1-propan(= 230.3) gives the corresponding acetate (= 15.3) when heated inacetic acid. If complete inversion had occurred, the optically pure acetatewould have had= 153.6. What percentage racemization and what percentageinversion occurred in this reaction?
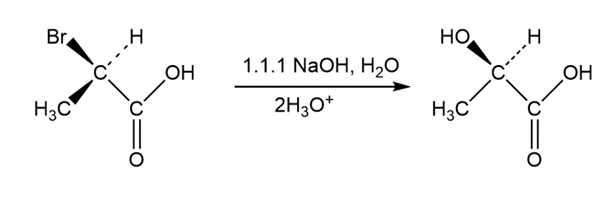
SN2 reactions take place with inversion of configuration, andSN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose a mechanism.
.
What do you think about this solution?
We value your feedback to improve our textbook solutions.