Chapter 11: Q11.73E (page 350)
Propose a structure for an alkyl halide that gives only (E)-3-methyl-2-phenyl-2-pentene on elimination. Make sure you indicate the stereochemistry.
Short Answer
 Formation of the desired product
Formation of the desired product
Learning Materials
Features
Discover
Chapter 11: Q11.73E (page 350)
Propose a structure for an alkyl halide that gives only (E)-3-methyl-2-phenyl-2-pentene on elimination. Make sure you indicate the stereochemistry.
 Formation of the desired product
Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
What product would you expect from a nucleophilic substitution reaction of (S) -2-bromohexane with acetate ion, CH3CO2-? Assume that inversion of configuration occurs, and show the stereochemistry of both the reactant and
product.
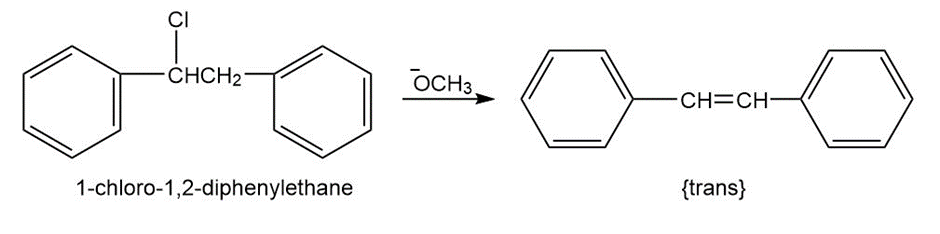
Question: 1-Chloro-1,2-diphenylethane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw Newman projections of the reactive conformations leading to both possible products, and suggest a reason why the trans alkene is the major product.
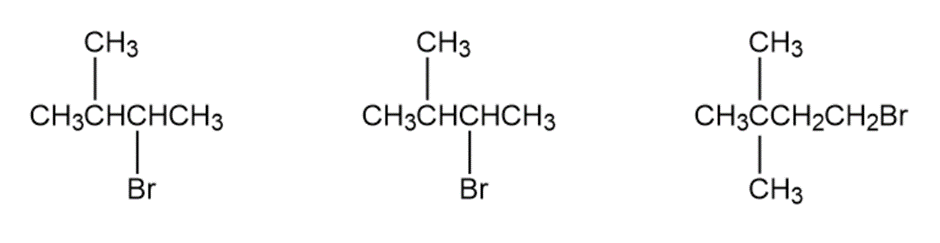
Question: Order each of the following sets of compounds with respect to reactivity:
b.
Question: What stereochemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1,2-dibromo-1,2-diphenylethane? Draw a Newman projection of the reacting conformation.
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.