Chapter 11: Q47cE (page 350)
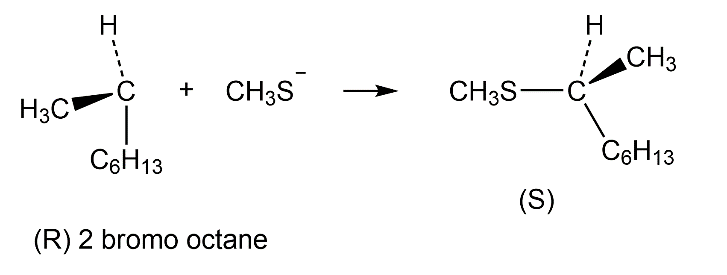
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
(c)
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 11: Q47cE (page 350)
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
(c)
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
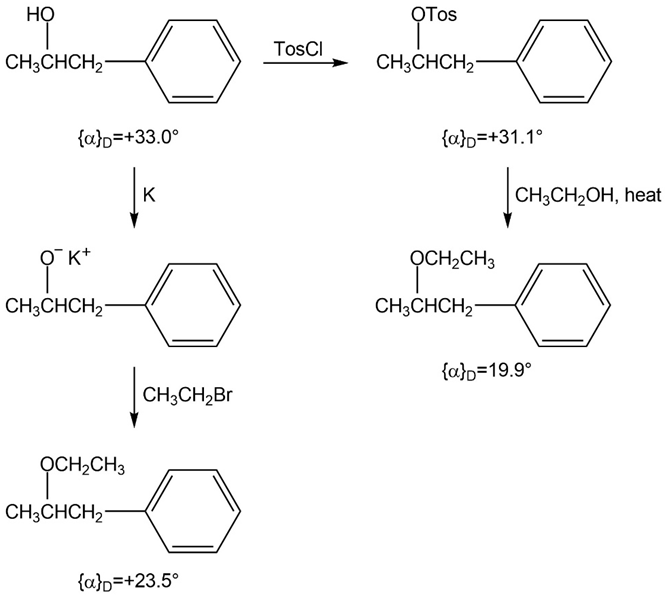
The following Walden cycle has been carried out. Explain the results, and indicate where Walden inversion occurs.
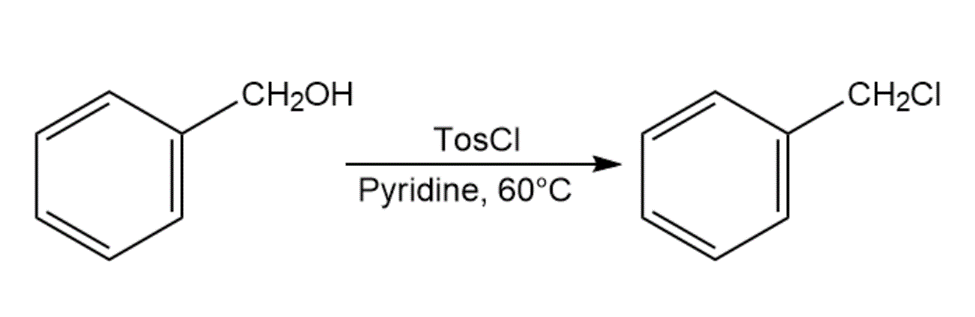
When primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at a higher temperature, an alkyl chloride is often formed. Explain.
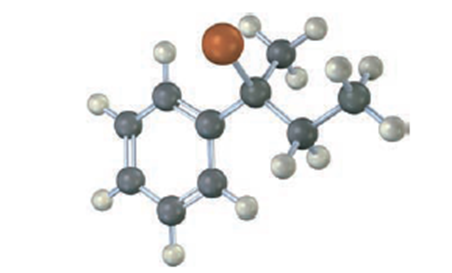
Assign configuration to the following substrate, and show the stereochemistry and identity of the product you would obtain by reaction with water (reddish-brown=Br):
Question: There are eight diastereomers of 1,2,3,4,5,6-hexachlorocyclohexane. Draw each in its more stable chair conformation. One isomer loses HCl in an E2 reaction nearly 1000 times more slowly than the others. Which isomer reacts so slowly, and why?
Which reaction in each of the following pairs would you expect to be faster?
(a) Thedisplacement by
What do you think about this solution?
We value your feedback to improve our textbook solutions.