Chapter 11: Q8 P. (page 326)
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereochemistry of both reactant and product
Short Answer

Learning Materials
Features
Discover
Chapter 11: Q8 P. (page 326)
What product(s) would you expect from reaction of (S)-3-chloro-3-methyloctane with acetic acid? Show the stereochemistry of both reactant and product

All the tools & learning materials you need for study success - in one app.
Get started for free
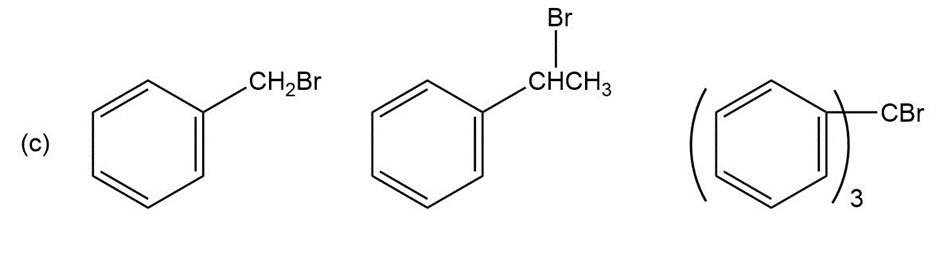
Question: Order each of the following sets of compounds with respect to reactivity:
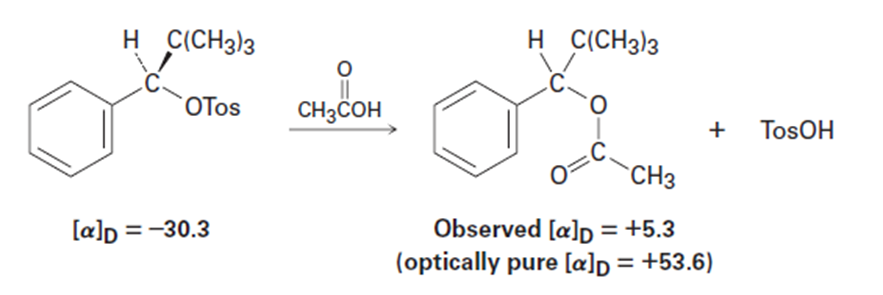
Among the many examples of reactions that occur with incomplete racemization, the optically pure tosylate of 2,2-dimethyl-1-phenyl-1-propan(= 230.3) gives the corresponding acetate (= 15.3) when heated inacetic acid. If complete inversion had occurred, the optically pure acetatewould have had= 153.6. What percentage racemization and what percentageinversion occurred in this reaction?
Which reactant in each of the following pairs is more nucleophilic? Explain.
a) NH2-or NH3
Question:What products would you expect from the reaction of 1-bromopropane with each of the following?
(f) Mg, then H2O
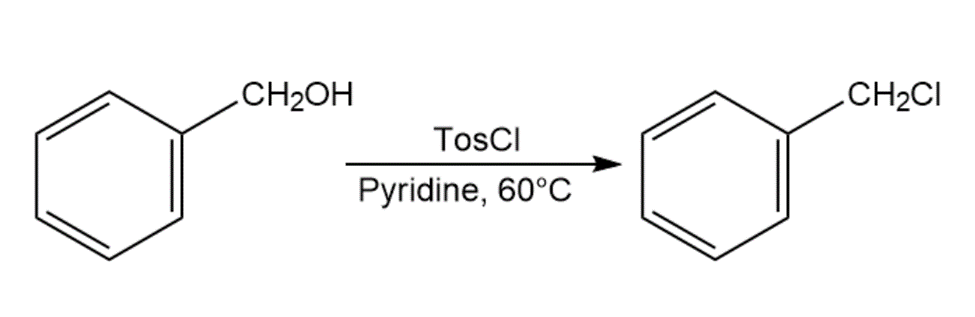
When primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at a higher temperature, an alkyl chloride is often formed. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.