Chapter 11: Q56C (page 350)
Question: Order each of the following sets of compounds with respect to reactivity:
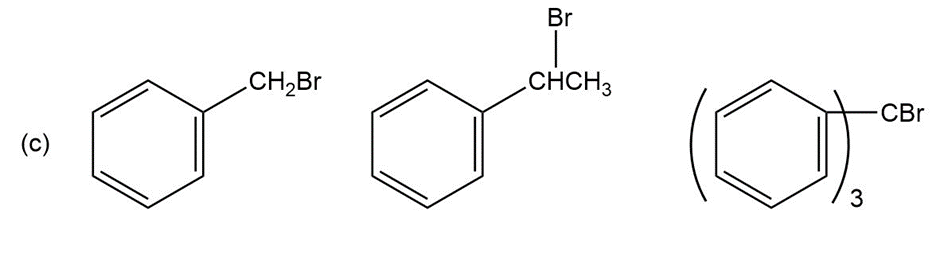
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 11: Q56C (page 350)
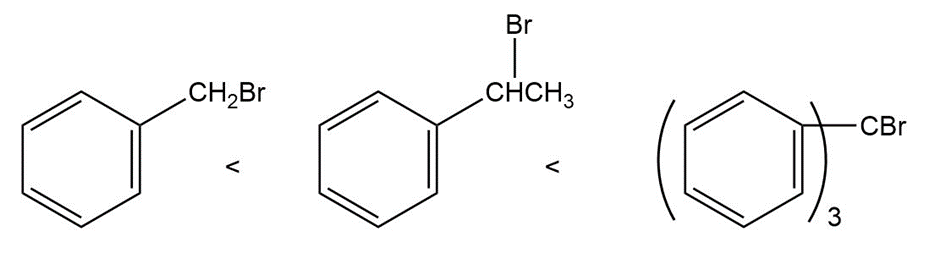
Question: Order each of the following sets of compounds with respect to reactivity:

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the product of each reaction below and indicate if the mechanism is likely to be
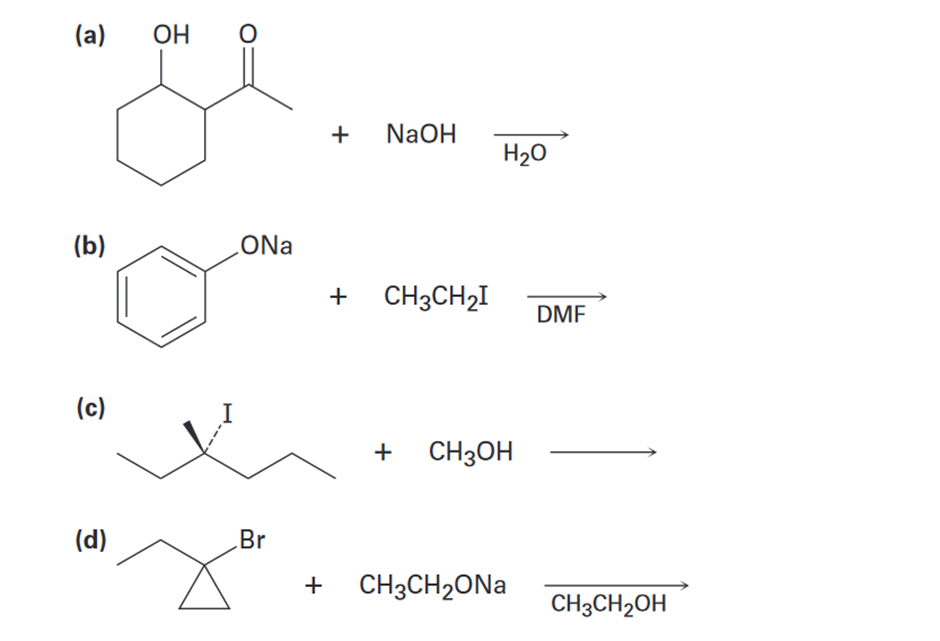
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
b)
Question: Order each of the following sets of compounds with respect to reactivity:
b. (CH3)3 CCI (CH3)3 CBr (CH3)3 COH
When primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at a higher temperature, an alkyl chloride is often formed. Explain.
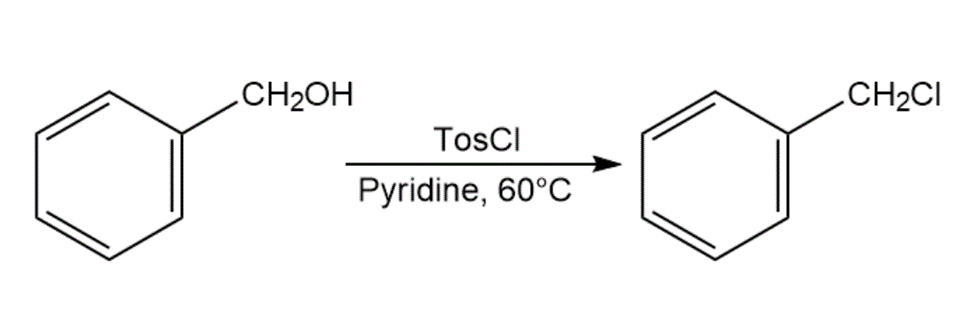
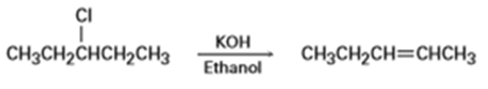
Which reaction in each of the following pairs would you expect to be faster?
(d) Thedisplacement byon bromomethane in benzene or in acetonitrile
What do you think about this solution?
We value your feedback to improve our textbook solutions.