Chapter 11: Q 11-11-20 P-b (page 347)
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
b)
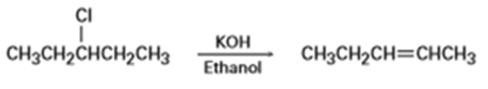
Short Answer
b) The given reaction is an E2 reaction
Learning Materials
Features
Discover
Chapter 11: Q 11-11-20 P-b (page 347)
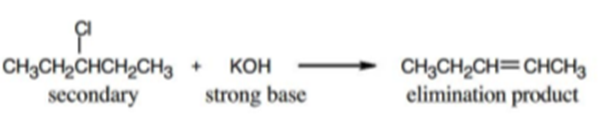
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB or E2:
b)

b) The given reaction is an E2 reaction

All the tools & learning materials you need for study success - in one app.
Get started for free
Which reactant in each of the following pairs is more nucleophilic? Explain.
a) BF3 or F-
Question: Order each of the following sets of compounds with respect to reactivity:
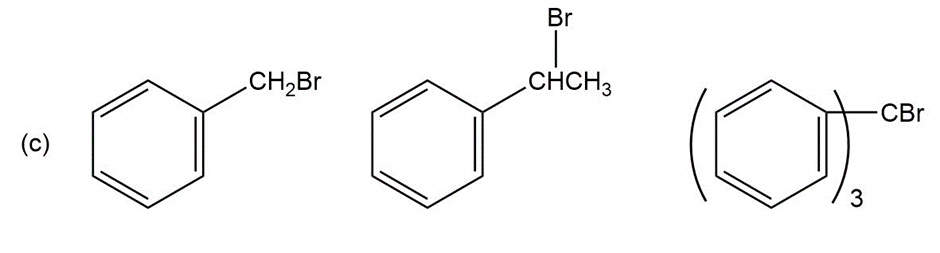
Question: Propose structures for compounds that fit the following descriptions:
(b) An organohalide that will not undergo nucleophilic substitution
Methyl esters () undergo a cleavage reaction to yield carboxylate ions plus iodomethane on heating with LiI in dimethylformamide:
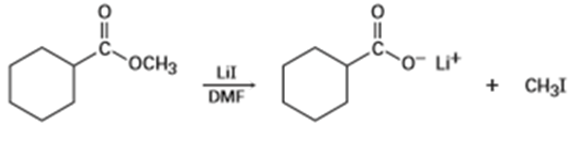
The following evidence has been obtained: (1) The reaction occurs much faster in DMF than in ethanol. (2) The corresponding ethyl ester () cleaves approximately 10 times more slowly than the methyl ester. Propose a mechanism for the reaction. What other kinds of experimental evidence could you gather to support your hypothesis?
Question: What stereochemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1,2-dibromo-1,2-diphenylethane? Draw a Newman projection of the reacting conformation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.