Chapter 11: 46cE (page 350)
Which reaction in each of the following pairs would you expect to be faster?
(c) Thedisplacement on 2-bromopropane byor by
Learning Materials
Features
Discover
Chapter 11: 46cE (page 350)
Which reaction in each of the following pairs would you expect to be faster?
(c) Thedisplacement on 2-bromopropane byor by
All the tools & learning materials you need for study success - in one app.
Get started for free
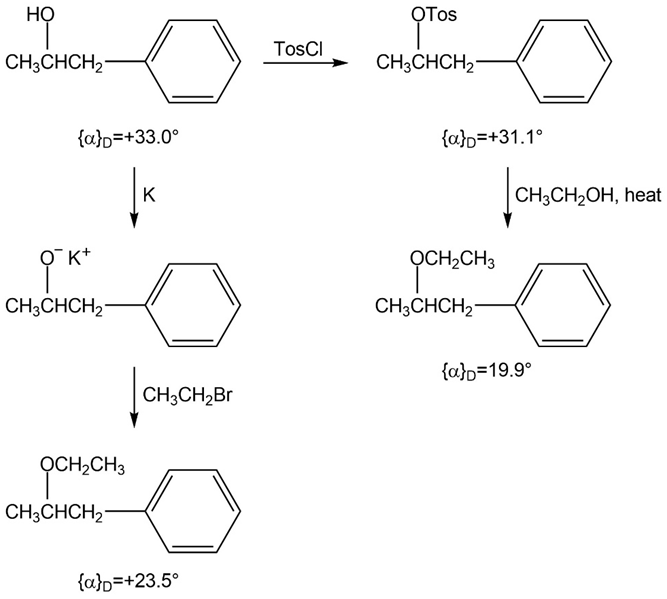
The following Walden cycle has been carried out. Explain the results, and indicate where Walden inversion occurs.
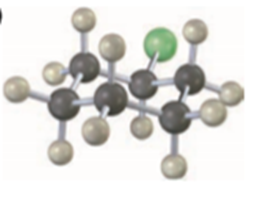
Question: Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane:
a)
Write the product you would expect from reaction of each of the following alkyl halides with (1)Na+-SCH3and (2)Na+-OH (green = Cl):
c)
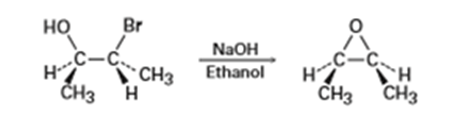
We saw in Section 8-7 that bromohydrins are converted into epoxides when treated with base. Propose a mechanism, using curved arrows to show the electron flow.
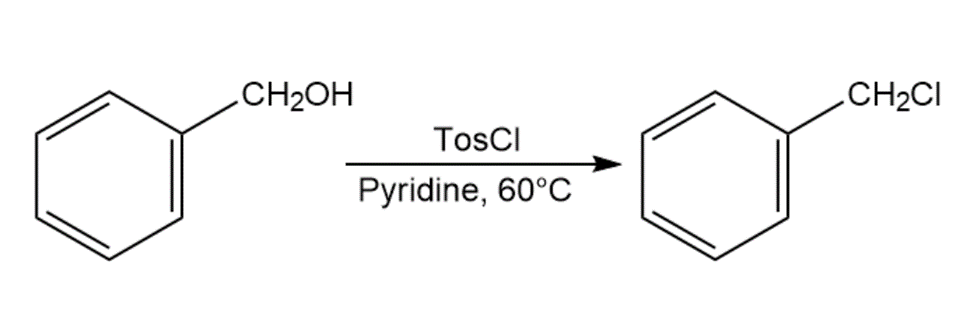
When primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out at a higher temperature, an alkyl chloride is often formed. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.