Chapter 18: Q20E (page 594)
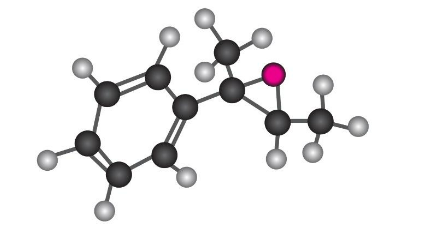
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q20E (page 594)
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr


All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following ethers? Use whichever method you think is more appropriate, Williamson synthesis or the alkoxymercuration reaction.
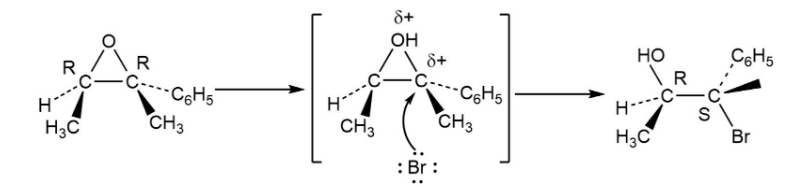
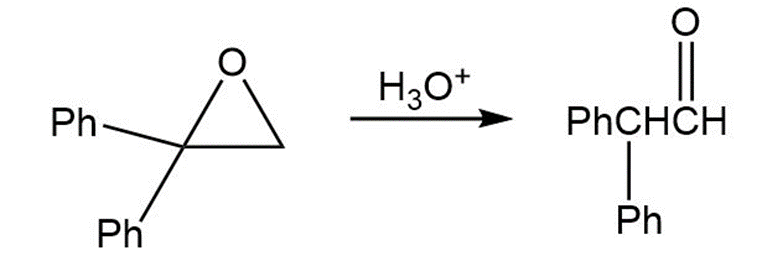
Treatment of 1,1-diphenyl-1,2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
Write the mechanism of the hydrolysis of cis-5,6-epoxydecane by reaction with aqueous acid. What is the stereochemistry of the product, assuming normal backside SN2 attack?
The red fox (Vulpesvulpes) uses a chemical communication system based on scent marks in urine. One component of fox urine is a sulfide whose mass spectrum has . IR spectroscopy shows an intense band at and NMR spectroscopy reveals the following peaks:
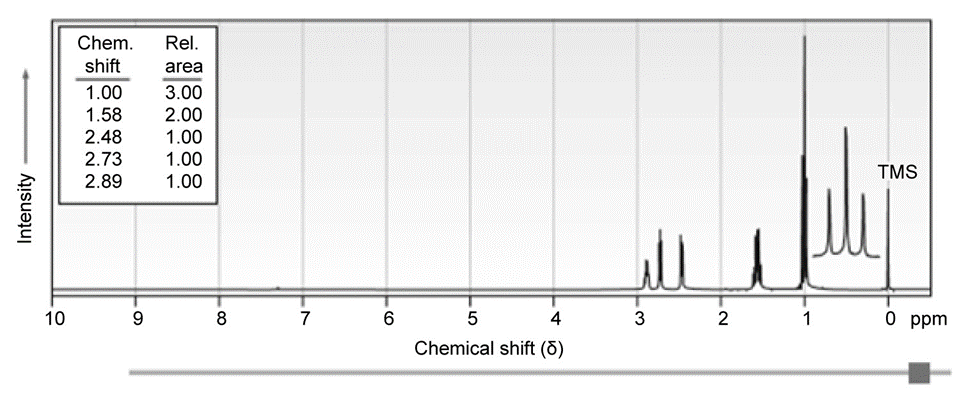
(, singlet); (, singlet); (, triplet, ); (, triplet, ); (, broad). Propose a structure consistent with these data. [Note: absorbs at ].The 1H NMR spectrum shown is that of a cyclic ether with the formula C4H8O. Propose a structure.
What do you think about this solution?
We value your feedback to improve our textbook solutions.