Chapter 18: Q19E (page 594)
Give IUPAC names for the following compounds (reddish brown 5 Br; yellow 5 S):
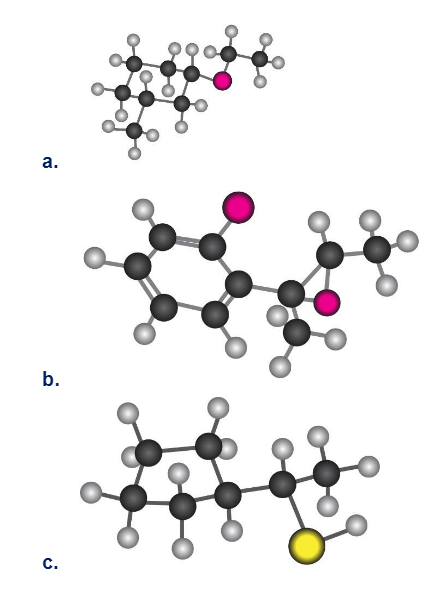
Short Answer
a. cis-1-ethoxy,3-methyl cyclohexane
b. E-2(o-bromo phenyl)-2,3-ethoxy butane
c. S-1-cyclopential ethane thiol
Learning Materials
Features
Discover
Chapter 18: Q19E (page 594)
Give IUPAC names for the following compounds (reddish brown 5 Br; yellow 5 S):

a. cis-1-ethoxy,3-methyl cyclohexane
b. E-2(o-bromo phenyl)-2,3-ethoxy butane
c. S-1-cyclopential ethane thiol
All the tools & learning materials you need for study success - in one app.
Get started for free
Reaction of cis-2-butene with m-chloroperoxybenzoic acid yields an epoxide different from that obtained by reaction of the trans isomer. Explain.
Rank the following halides in order of their reactivity in Williamson synthesis:
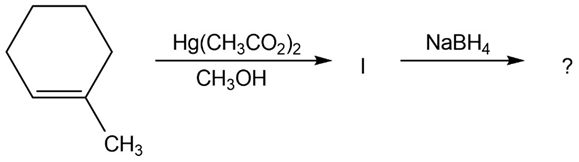
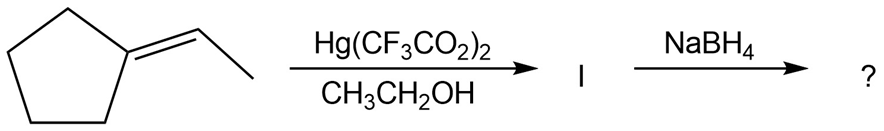
The alkoxymercuration of alkenes involves the formation of an organomercury intermediate (I), which is reduced with NaBH4 to give an etherproduct. For each reaction below, predict the ether product and provide the mechanism formation.
(a)
(b)
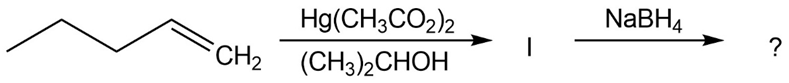
(c)
How would you prepare the following compounds from 1-phenylethanol?
(a)Methyl 1-phenylethyl ether (b) Phenylepoxyethane
(c)tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
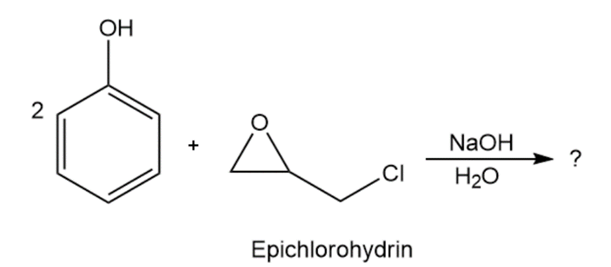
In the formation of the prepolymer used to make epoxy resins, a bisphenol reacts with epichlorohydrin in the presence of a base. Show the product and mechanism when two moles of phenol react with epichlorohydrin.
What do you think about this solution?
We value your feedback to improve our textbook solutions.