Chapter 18: Q41E (page 594)
How would you prepare the following compounds from 1-phenylethanol?
(a)Methyl 1-phenylethyl ether (b) Phenylepoxyethane
(c)tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
Short Answer
(a)
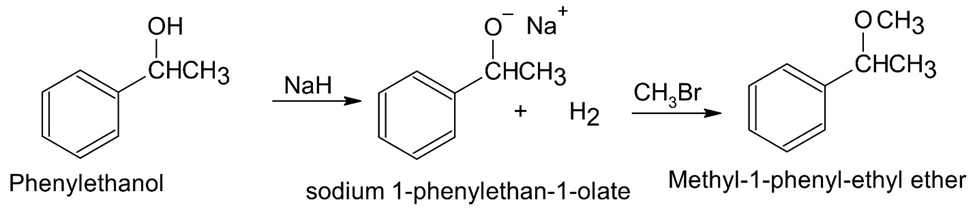
(b)

(c)

(d)
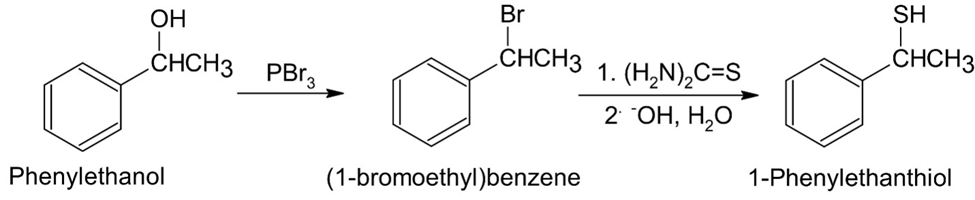
Learning Materials
Features
Discover
Chapter 18: Q41E (page 594)
How would you prepare the following compounds from 1-phenylethanol?
(a)Methyl 1-phenylethyl ether (b) Phenylepoxyethane
(c)tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following ethers using a Williamson synthesis?
Rank the following halides in order of their reactivity in Williamson synthesis:
Meerwein’s reagent, triethyloxoniumtetrafluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein’s reagent with cyclohexanol, and account for the fact that trialkyloxonium salts are much more reactive alkylating agents than alkyl iodides.
Meerwein’s reagent
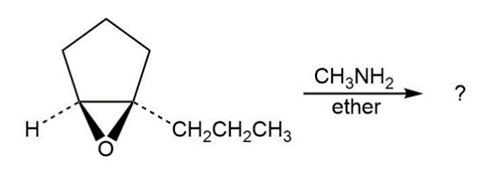
Predict the product(s) and provide the mechanism for each reaction below. What do the mechanisms have in common?
a.
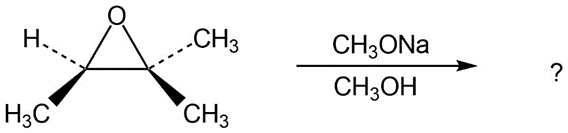
b.
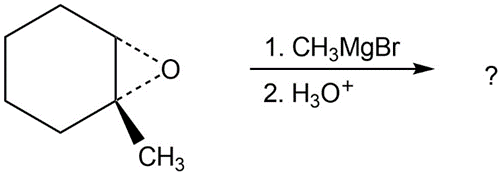
c.
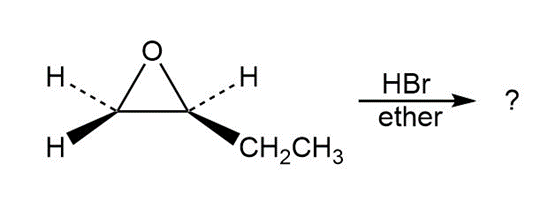
d.
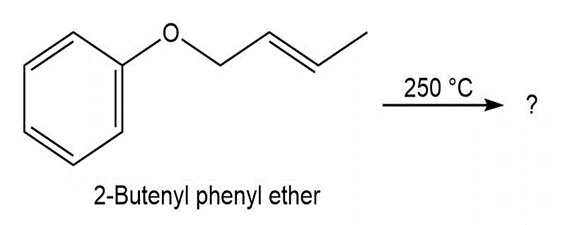
What product would you expect from Claisen rearrangement of 2-butenyl phenyl ether?
What do you think about this solution?
We value your feedback to improve our textbook solutions.