Chapter 18: Q40E (page 594)
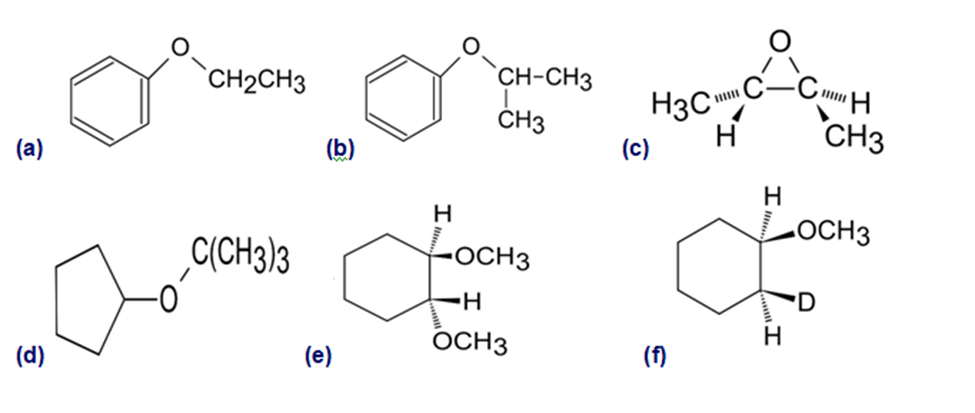
How would you prepare the following ethers?
Short Answer
(a)
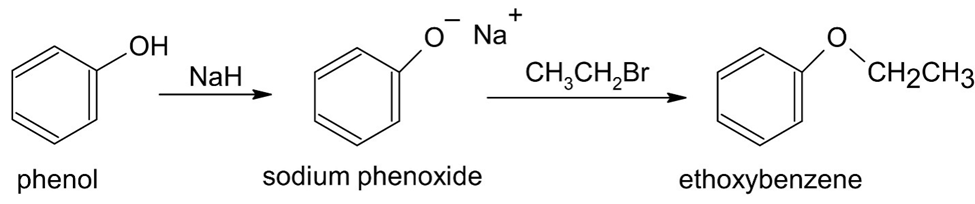
(b)
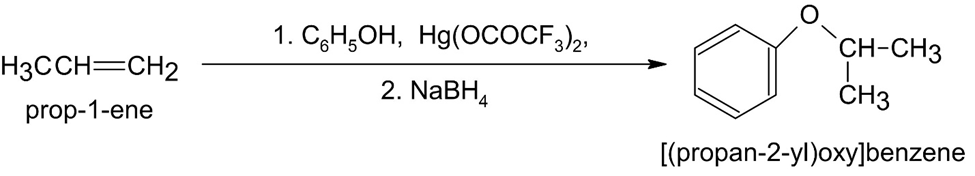
(c)
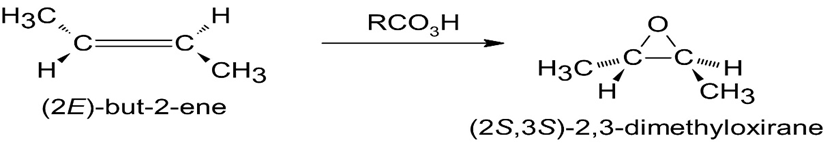
(d)
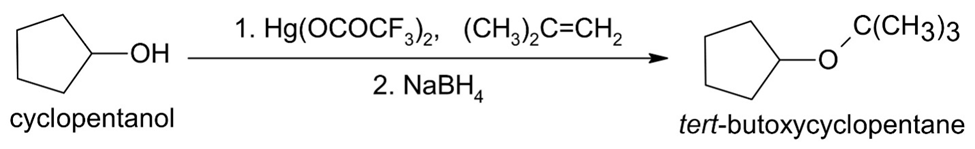
(e)
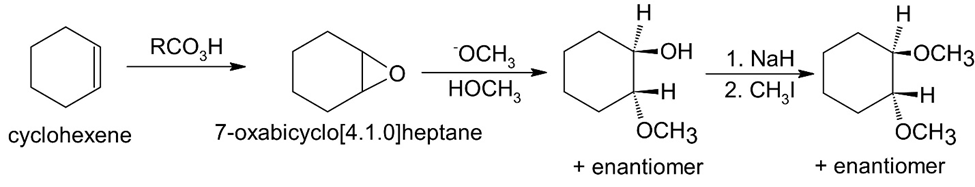
(f)
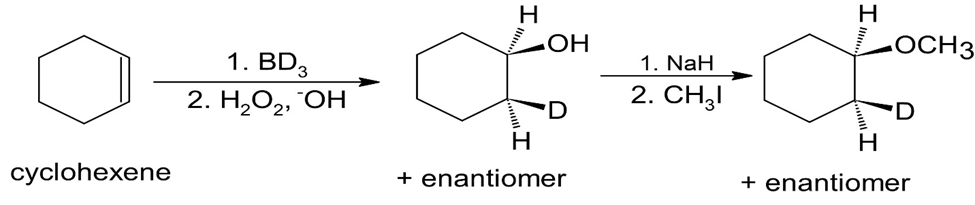
Learning Materials
Features
Discover
Chapter 18: Q40E (page 594)
How would you prepare the following ethers?

(a)

(b)
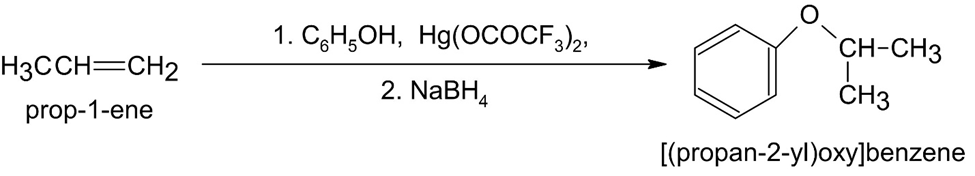
(c)

(d)
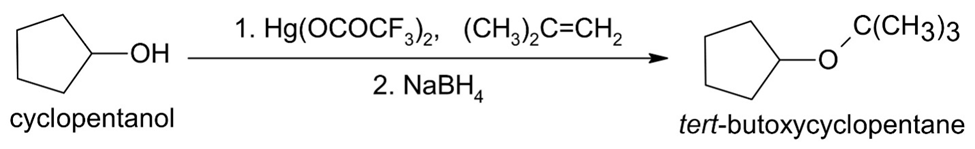
(e)

(f)

All the tools & learning materials you need for study success - in one app.
Get started for free
Write the mechanism of the hydrolysis of cis-5,6-epoxydecane by reaction with aqueous acid. What is the stereochemistry of the product, assuming normal backside SN2 attack?
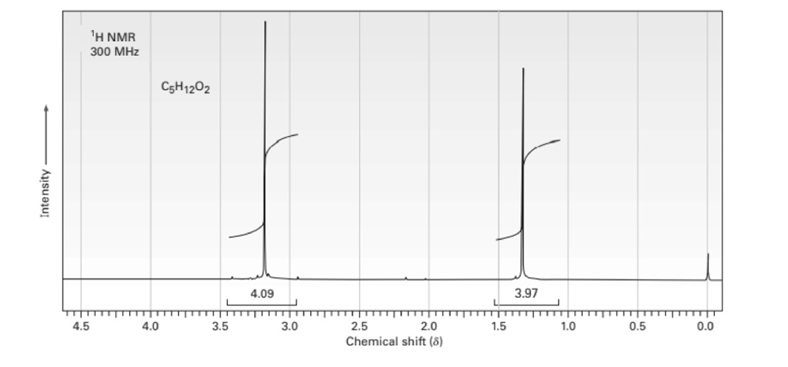
Propose structures for compounds that have the following 1H NMR spectra:
(a) C5H12S (An –SH proton absorbs near 1.6)
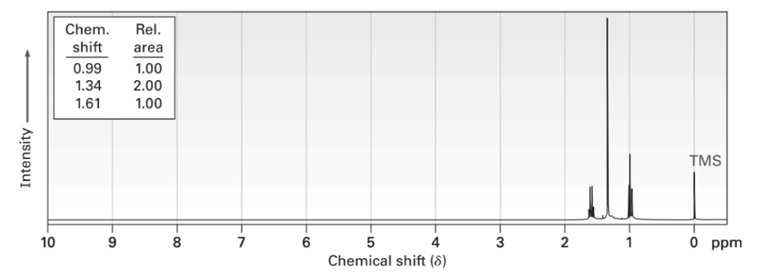
(b)C9H11BRO
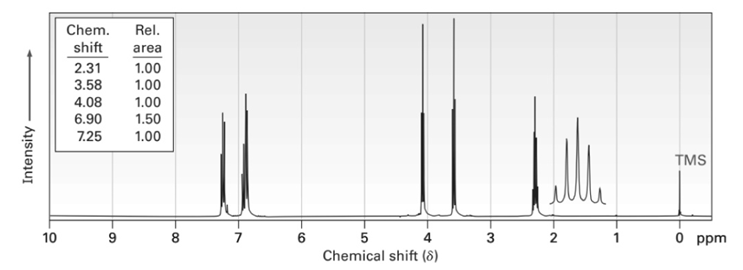
(c)C5H12O2
In nature, the enzyme chorismate mutase catalyses a Claisen rearrangement of chorismate that involves both the terminal double bond and the double bond with the highlighted carbon. What is the structure of prephenate, the biological precursor to the amino acids phenylalanine and tyrosine?
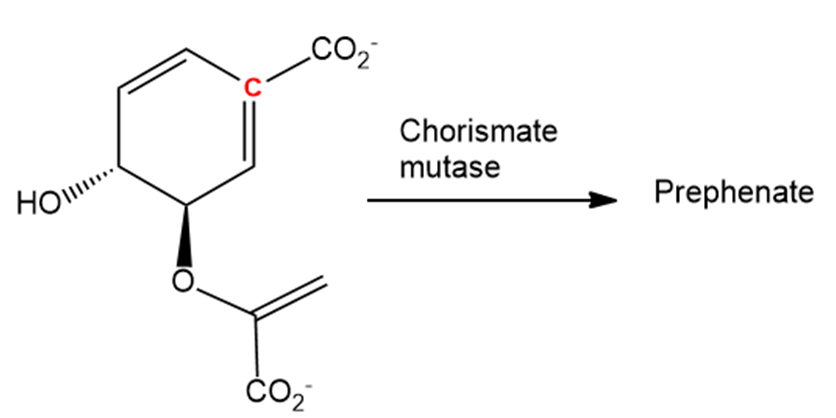
Chorismate
Meerwein’s reagent, triethyloxoniumtetrafluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein’s reagent with cyclohexanol, and account for the fact that trialkyloxonium salts are much more reactive alkylating agents than alkyl iodides.
Meerwein’s reagent
Predict the products of the following ether cleavage reactions:
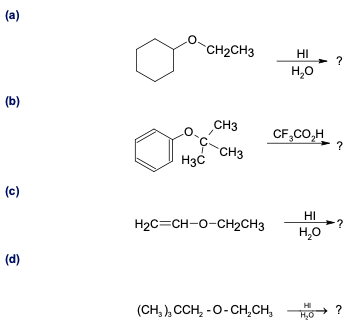
What do you think about this solution?
We value your feedback to improve our textbook solutions.