Chapter 18: Q31E (page 594)
Treatment of 1,1-diphenyl-1,2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
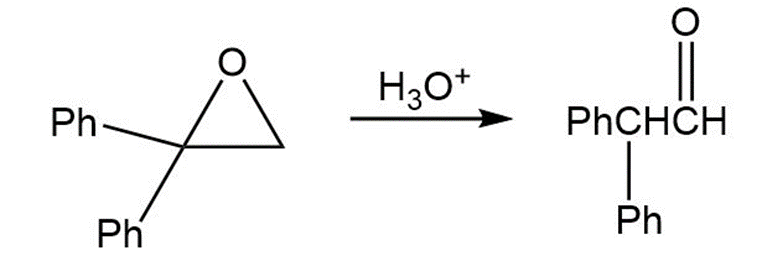
Short Answer
Mechanism followed as:
Learning Materials
Features
Discover
Chapter 18: Q31E (page 594)
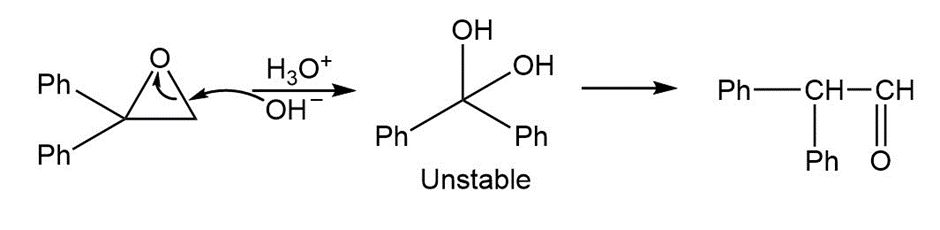
Treatment of 1,1-diphenyl-1,2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.

Mechanism followed as:

All the tools & learning materials you need for study success - in one app.
Get started for free
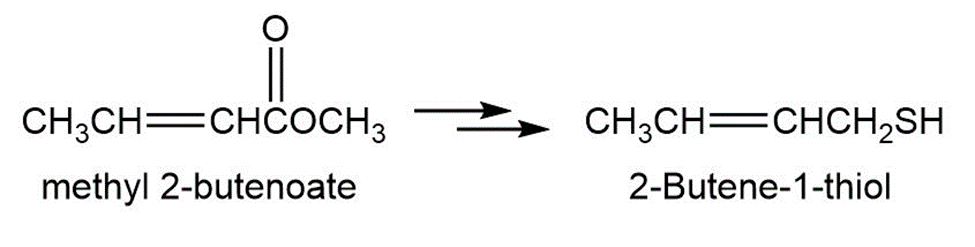
2-Butene-1-thiol is one component of skunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene?
How would you prepare the following ethers? Use whichever method you think is more appropriate, Williamson synthesis or the alkoxymercuration reaction.
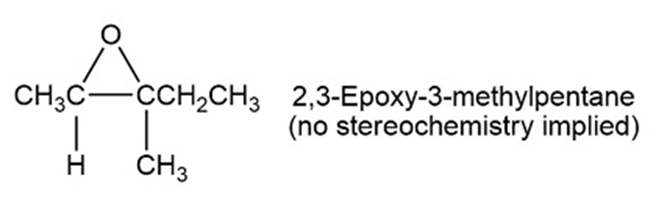
Imagine that you have created (2R, 3R)-2,3-epoxy-3-methylpentane with aqueous acid to carry out a ring opening reaction.
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
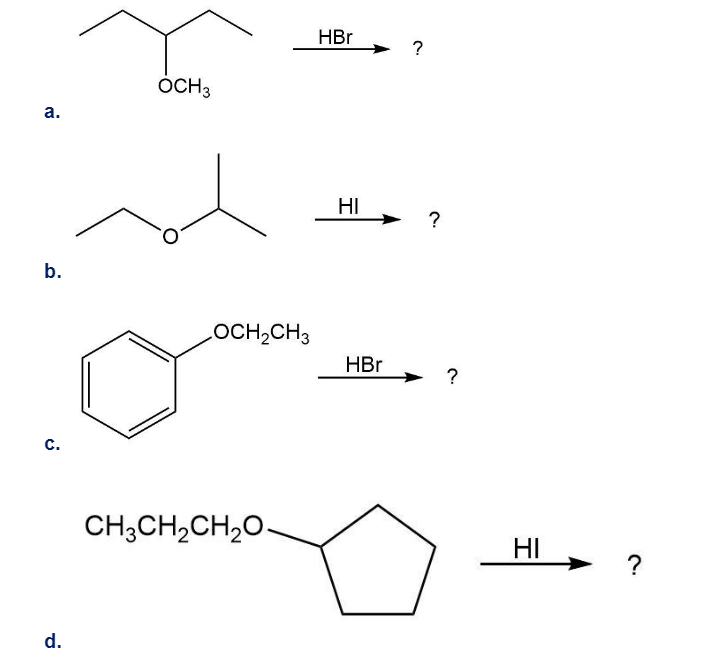
Why are HI and HBr more effective than HCl in cleaving ethers? (See Section 11-3).
What do you think about this solution?
We value your feedback to improve our textbook solutions.