Chapter 18: Q20-E (page 594)
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr
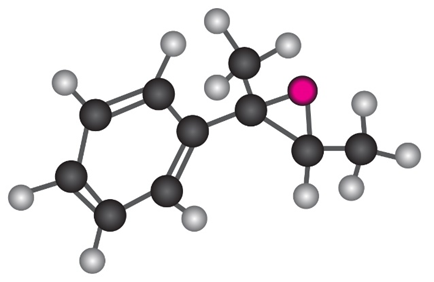
Short Answer
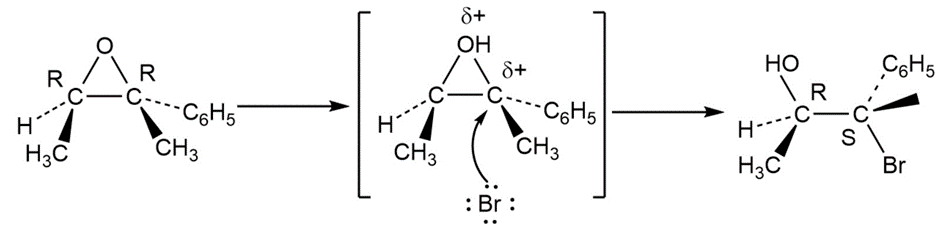
Learning Materials
Features
Discover
Chapter 18: Q20-E (page 594)
Show the product, including stereochemistry, that would result from reaction of the following epoxide with HBr


All the tools & learning materials you need for study success - in one app.
Get started for free
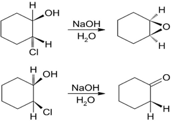
Treatment of trans-2-chlorocyclohexanol with NaOH yields 1,2-epoxycyclohexane,but reaction of the cis isomer under the same conditionsyields cyclohexanone. Propose mechanisms for both reactions, andexplain why the different results are obtained.
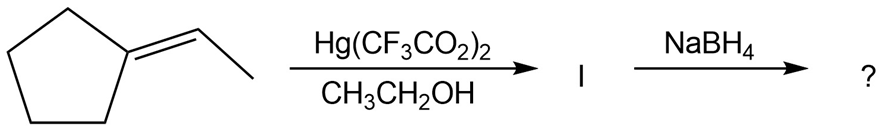
The alkoxymercuration of alkenes involves the formation of an organomercury intermediate (I), which is reduced with NaBH4 to give an etherproduct. For each reaction below, predict the ether product and provide the mechanism formation.
(a)
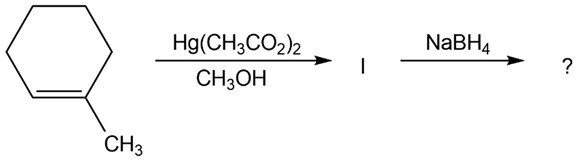
(b)
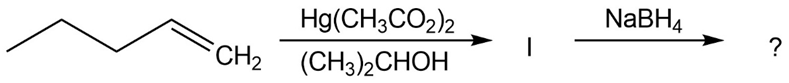
(c)
How could you prepare benzyl phenyl ether from benzene and phenol? More than one step is required.
Predict the products of the following reactions:
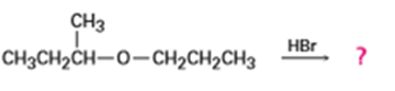
Review the mechanism of oxymercuration shown in Figure 8-3 on page 230, and then write the mechanism of the alkoxymercuration reaction of 1-methyl-cyclopentene with ethanol. Use curved arrows to show the electron flow in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.