Chapter 15: Benzene and Aromaticity
Q21P
Draw and name all possible aromatic compounds with the formula C7H7Cl.
Q22P
Draw and name all possible aromatic compounds with the formula C8H9Br.
Q41E
Propose structures for compounds that fit the following descriptions:
(a) NMR: (4 H, broad singlet);(4 H, quartet, J = 7 Hz); (6 H, triplet, J =7 Hz) IR:.
(b)NMR:(4 H, broad singlet);(1 H, septet, J = 8 Hz);(3 H, singlet);(6 H, doublet, J = 8 Hz) IR:.
Q42E
On reaction with acid, 4-pyrone is protonated on the carbonyl-group oxygen to give a stable cationic product. Using resonance structures and the Hückel’s rule, explain why the protonated product is so stable.
 4-Pyrone
4-Pyrone
Q43E
Bextra, a COX-2 inhibitor once used in the treatment of arthritis, contains an isoxazole ring. Why is the ring aromatic?
 Bextra
Bextra
Q44E
N-Phenylsydnone, so-named because it was first studied at the University of Sydney, Australia, behaves like a typical aromatic molecule. Explain, using the Hückel’s rule .
 N-phenylsydnone
N-phenylsydnone
Q45E
Show the relative energy levels of the seven p molecular orbitals of the cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the aromaticity of the cycloheptatrienyl cation.
Q46E
1-Phenyl-2-butene has an ultraviolet absorption (). On treatment with a small amount of strong acid, isomerization occurs and a new substance with () is formed. Propose a structure for this isomer, and suggest a mechanism for its formation.
Q47E
7 Propose structures for aromatic compounds that have the following NMR spectra:
(a)
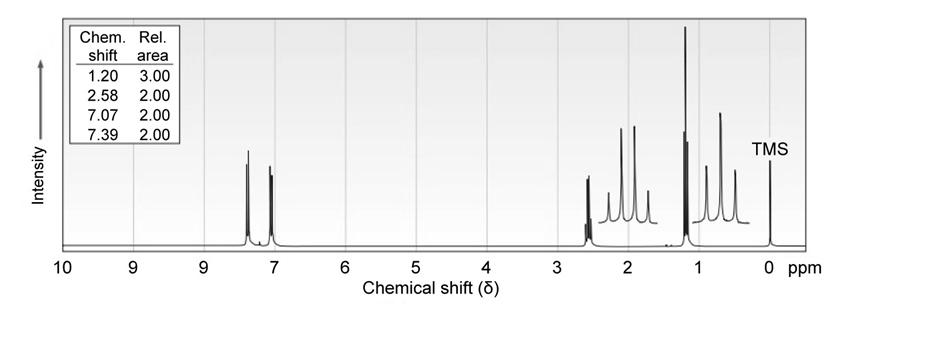
b)
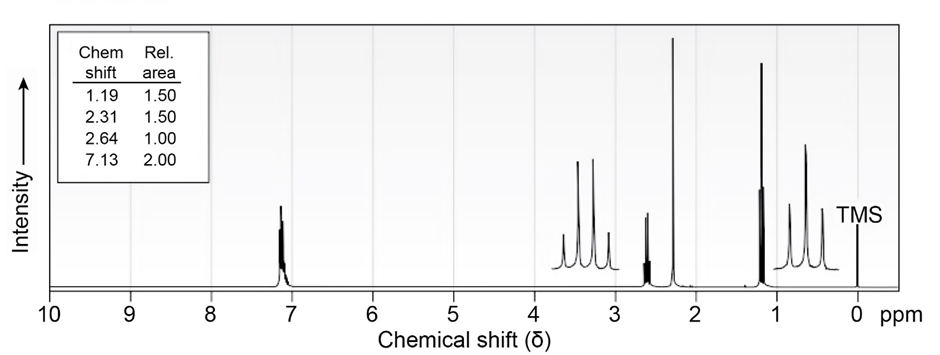
c)
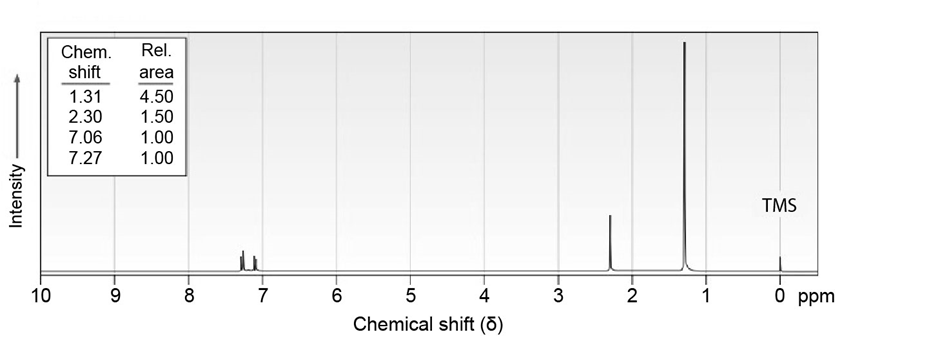
Q48E
Propose a structure for a molecule that has the following NMR spectrum and has IR absorptions at 700, 740, and :