Chapter 6: 6-35d (page 181)
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:
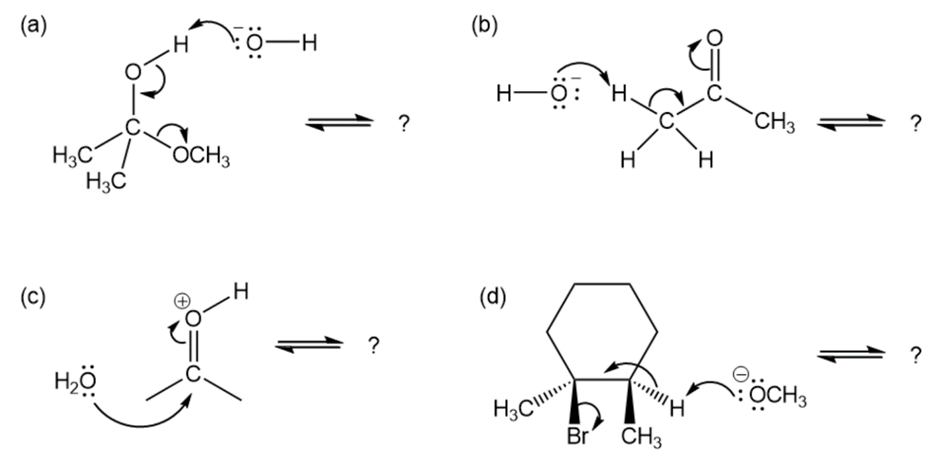
Short Answer
(d)
Formation of compound
Learning Materials
Features
Discover
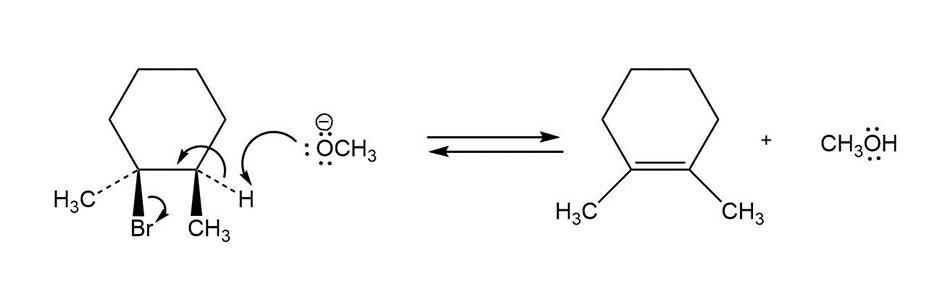
Chapter 6: 6-35d (page 181)
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:

(d)

Formation of compound
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the energy difference between the axial and equatorial conformations of cyclohexanol (hydroxycyclohexane)?
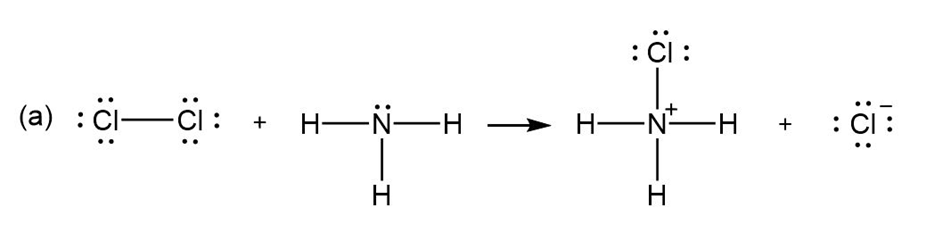
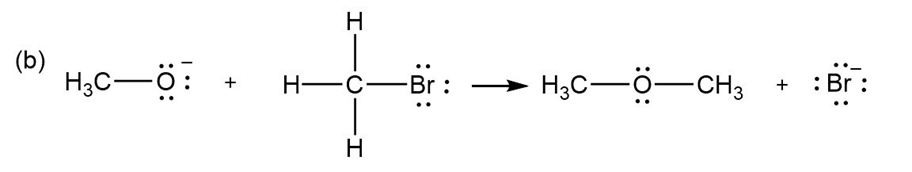
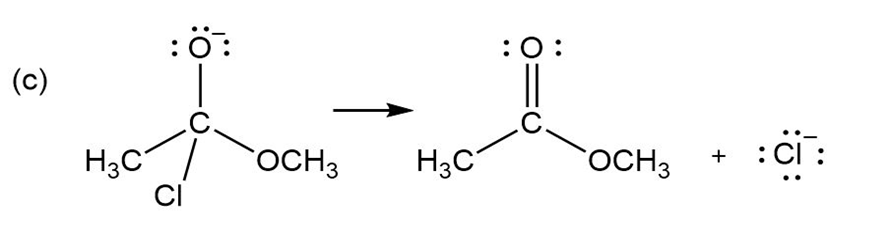
For each reaction below identify the electrophile and the nucleophile
(a)
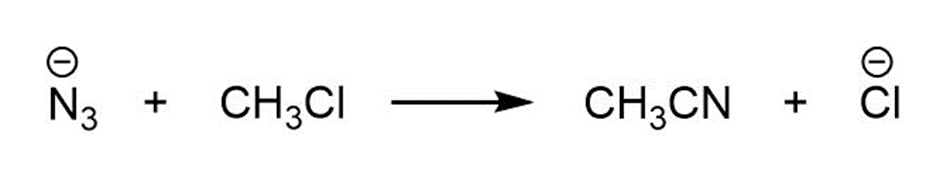
(b)
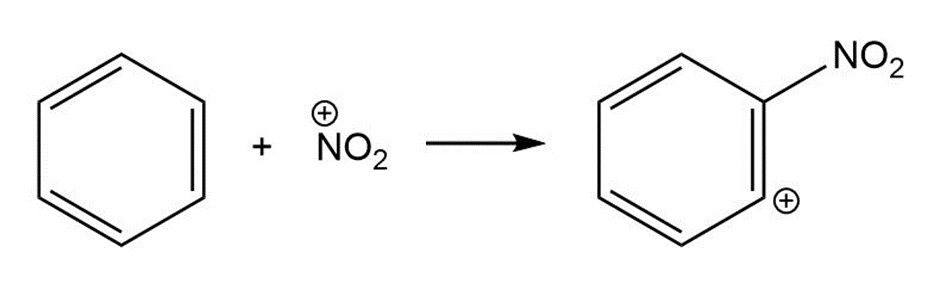
(c)
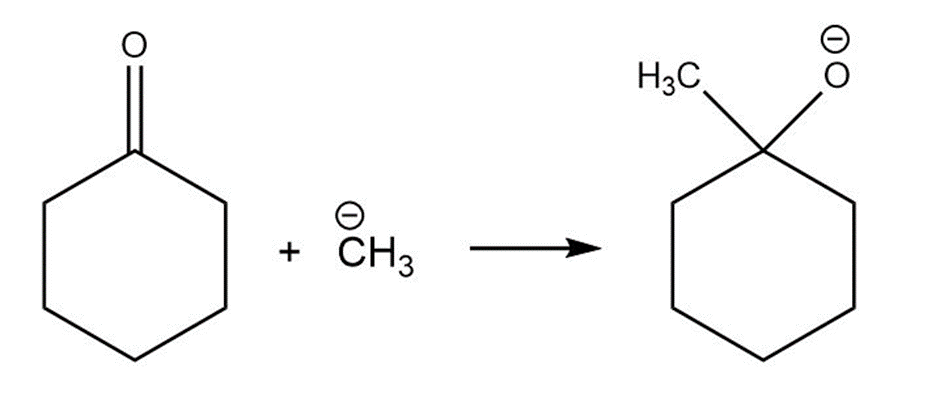
Add curved arrows to the following polar reactions to indicate the flow of electrons in each:
When a mixture of methane and chlorine is irradiated, a reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?
What do you think about this solution?
We value your feedback to improve our textbook solutions.