Chapter 6: 6-33-E-b (page 181)
For each reaction below identify the electrophile and the nucleophile
(a)
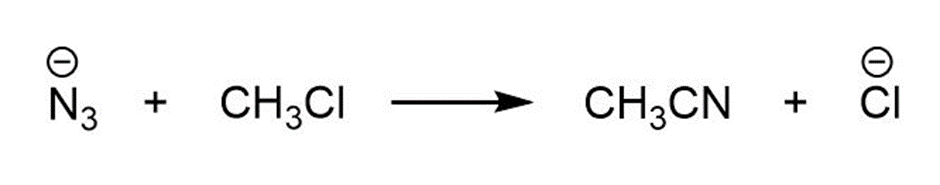
(b)
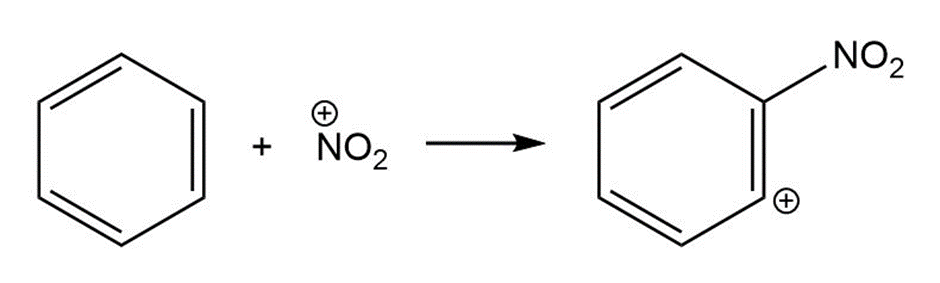
(c)
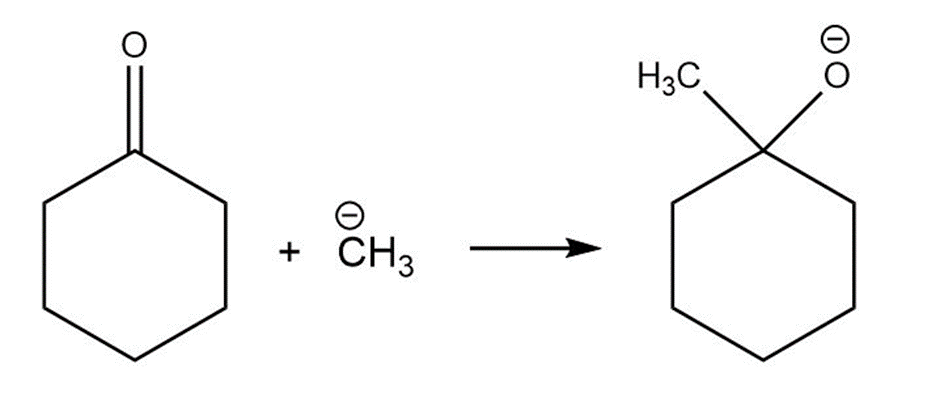
Short Answer
(b)
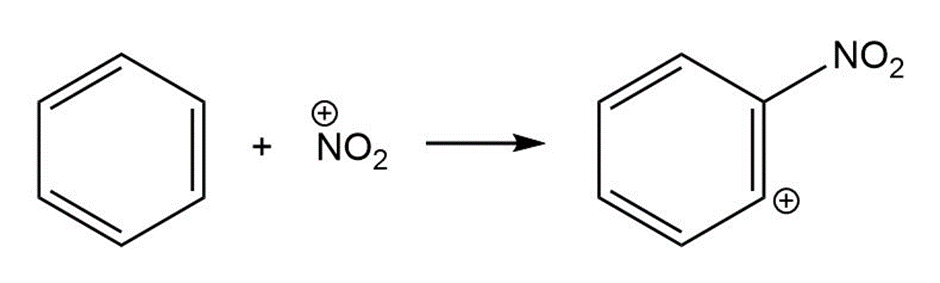
Nucleophile Electrophile
Learning Materials
Features
Discover
Chapter 6: 6-33-E-b (page 181)
For each reaction below identify the electrophile and the nucleophile
(a)

(b)

(c)

(b)

Nucleophile Electrophile
All the tools & learning materials you need for study success - in one app.
Get started for free
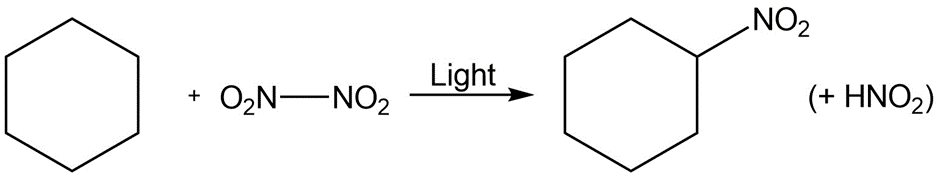
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.

b.
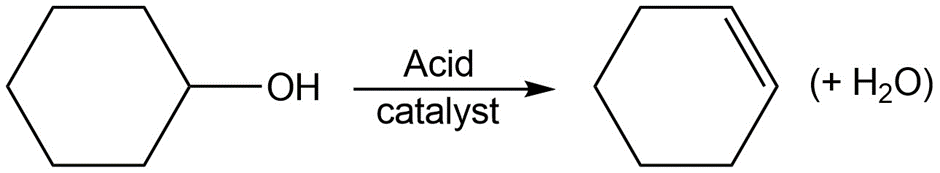
c.
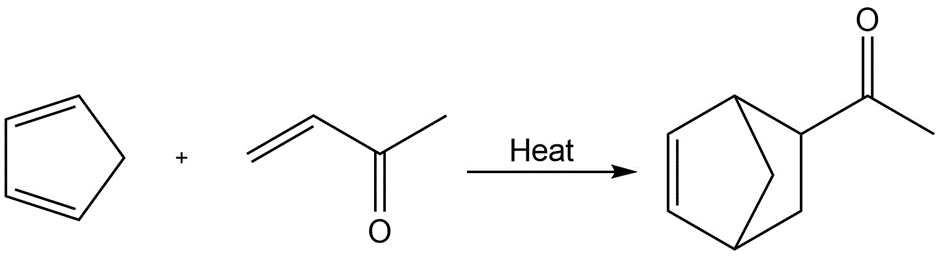
d.
Look at the following energy diagram:
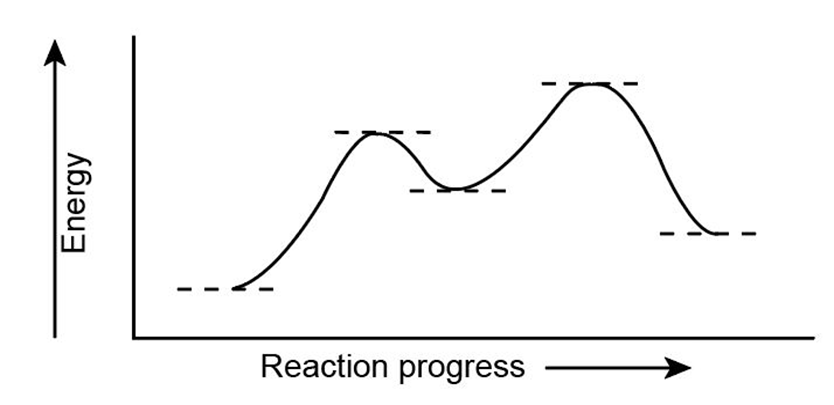
(a) Is for the reaction positive or negative? Label it on the diagram.
(b) How many steps are involved in the reaction?
(c) How many transition states are there? Label them on the diagram.
Radical chlorination of pentane is a poor way to prepare 1-chloropentane, but radical chlorination of neopentane,, is a good way to prepare neopentyl chloride,. Explain.
An electrostatic potential map of boron trifluoride is shown. Is likely to be a nucleophile or an electrophile? Draw a Lewis structure for , and explain your answer.
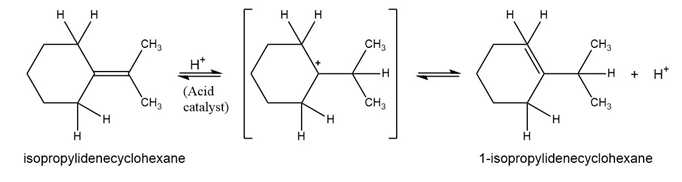
Add curved arrows to the mechanism shown in Problem 6-25 to indicate the electron movement in each step.
What do you think about this solution?
We value your feedback to improve our textbook solutions.