Chapter 6: 6-31D (page 181)
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.

b.
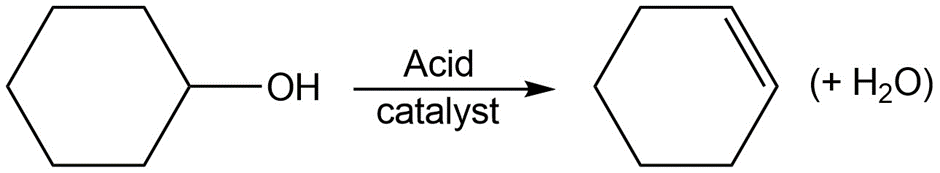
c.
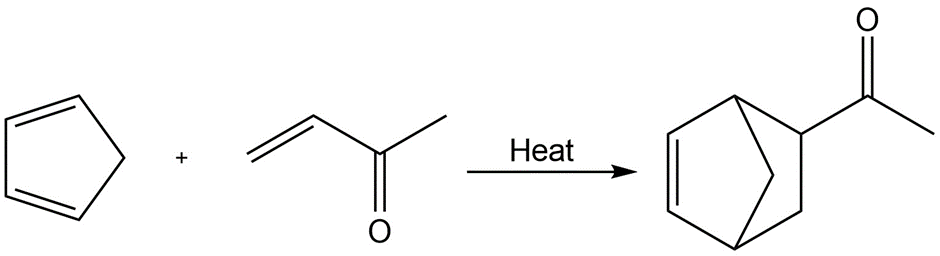
d.
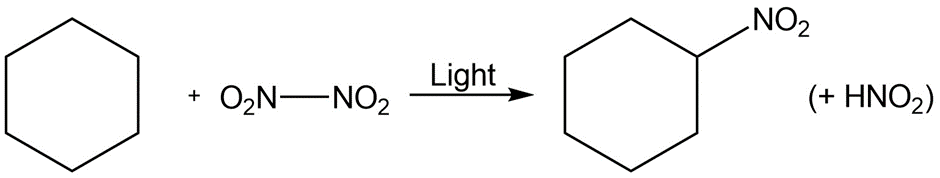
Short Answer
d.
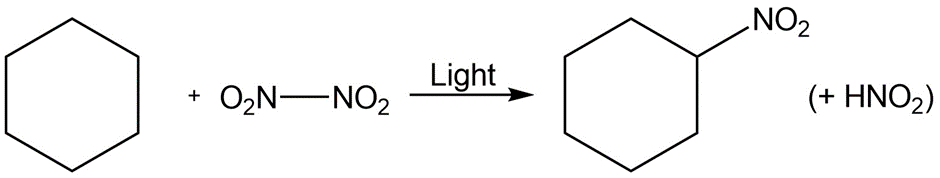
Substitution reaction
Learning Materials
Features
Discover
Chapter 6: 6-31D (page 181)
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.

b.

c.

d.

d.

Substitution reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement:
1.
2.
3.
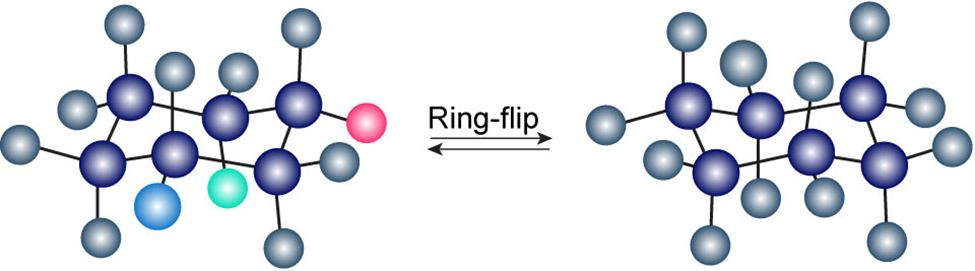
Question: Identify each of the colored positions—red, blue, and green—as axial or equatorial. Then carry out a ring-flip, and show the new positions occupied by each color.
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters:
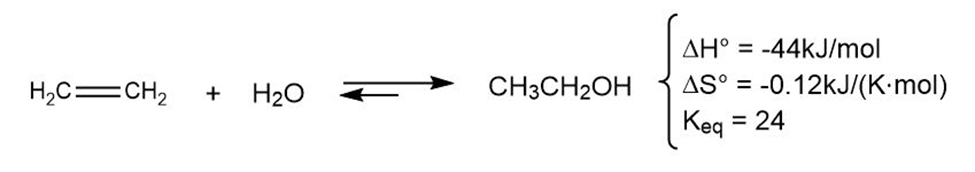
(a) Is the reaction exothermic or endothermic?
(b) Is the reaction favorable (spontaneous) or unfavorable (nonspontaneous) at room temperature (298 K)?
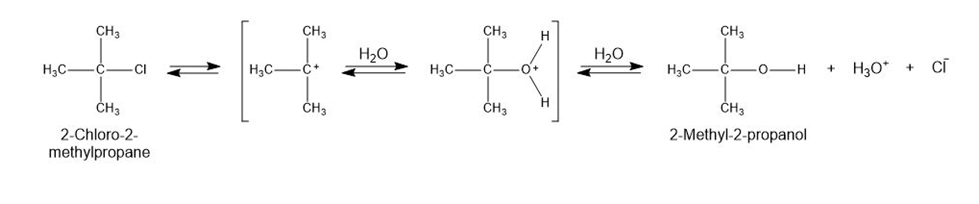
2-Chloro-2-methylpropane reacts with water in three steps to yield 2-methyl-2-propanol. The first step is slower than the second, which in turn is much slower than the third. The reaction takes place slowly at room temperature, and the equilibrium constant is approximately 1.
Draw the more stable chair conformation of the following molecules, and estimate the amount of strain in each:
What do you think about this solution?
We value your feedback to improve our textbook solutions.