Chapter 6: 6-31c (page 181)
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.
b.
c.
d.
Short Answer
c.
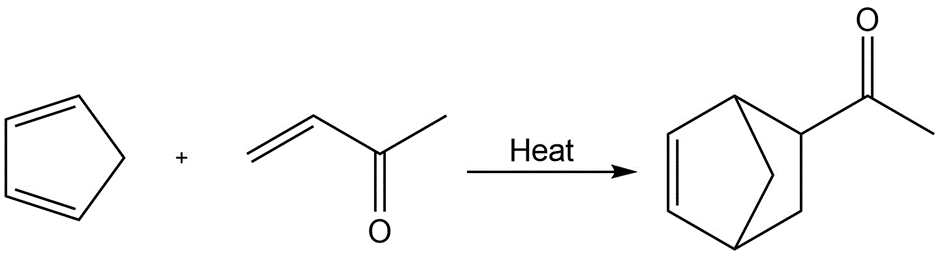
Addition reaction
Learning Materials
Features
Discover
Chapter 6: 6-31c (page 181)
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.
b.
c.
d.
c.

Addition reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Radical chlorination of pentane is a poor way to prepare 1-chloropentane, but radical chlorination of neopentane,, is a good way to prepare neopentyl chloride,. Explain.
What product would you expect from the reaction of cyclohexene with ? With ?
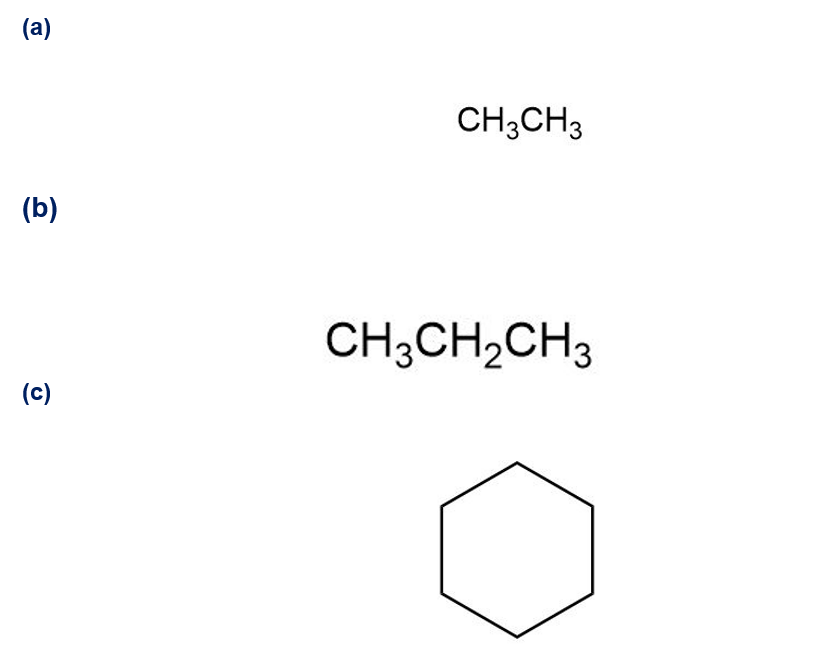
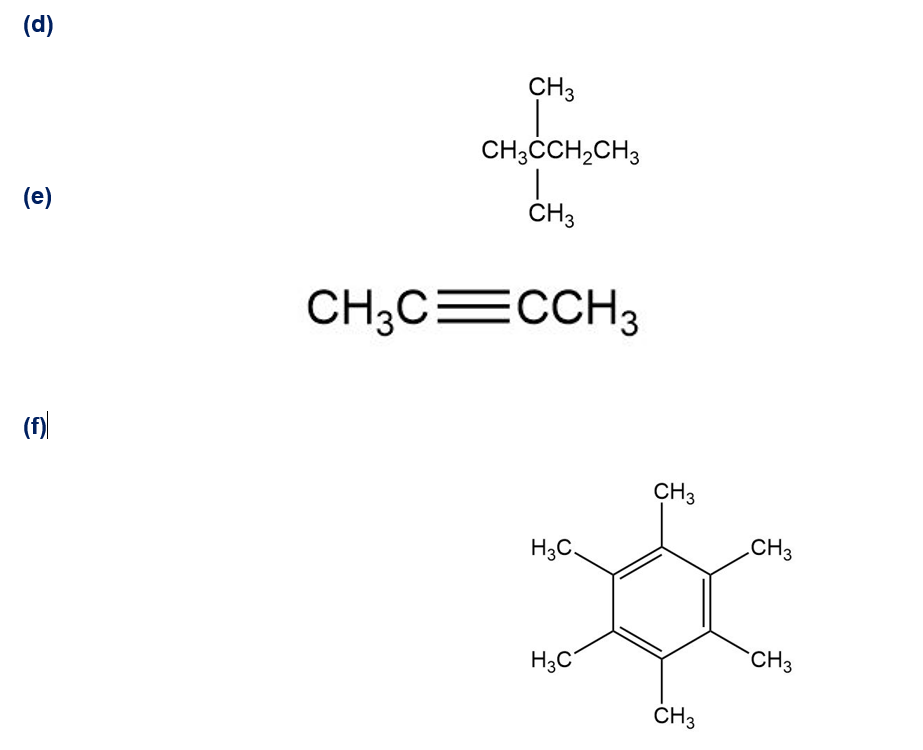
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination give a single monochloro product?
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step.
Add curved arrows to the mechanism shown in Problem 6-25 to indicate the electron movement in each step.
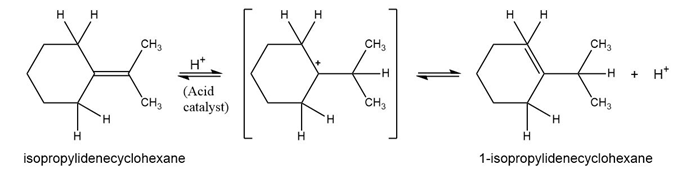
What do you think about this solution?
We value your feedback to improve our textbook solutions.