Chapter 6: 6-1 (page 151)
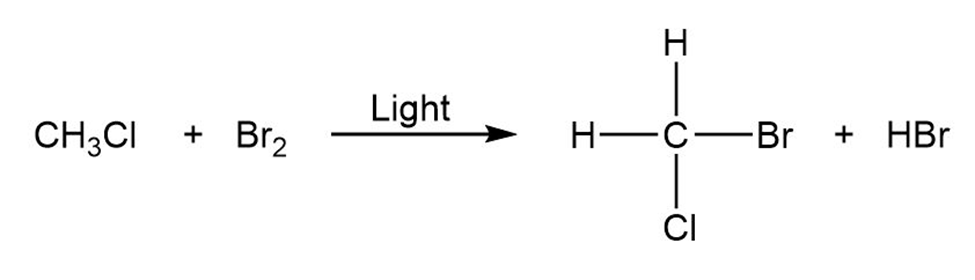
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement:
1.
2.
3.
Short Answer
- Substitution reaction
- Elimination reaction
- Addition reaction
Learning Materials
Features
Discover
Chapter 6: 6-1 (page 151)
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement:
1.
2.
3.
All the tools & learning materials you need for study success - in one app.
Get started for free
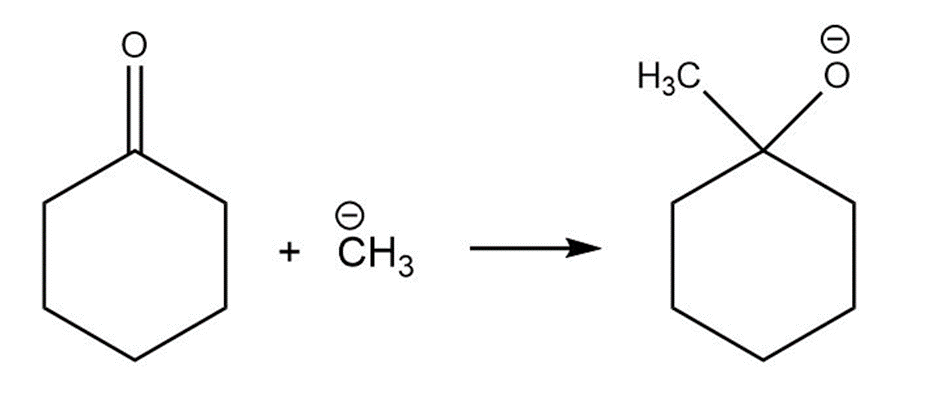
For each reaction below identify the electrophile and the nucleophile
(a)
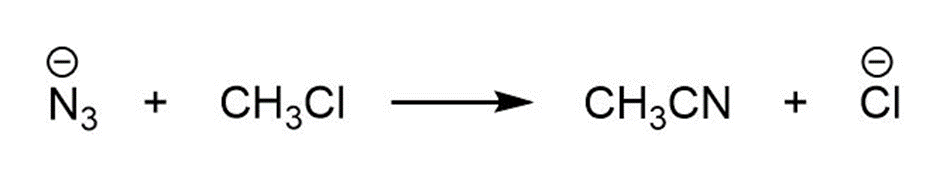
(b)

(c)
Draw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.
(a)
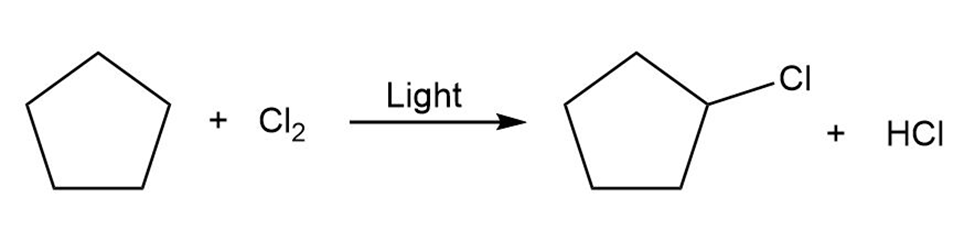
b)
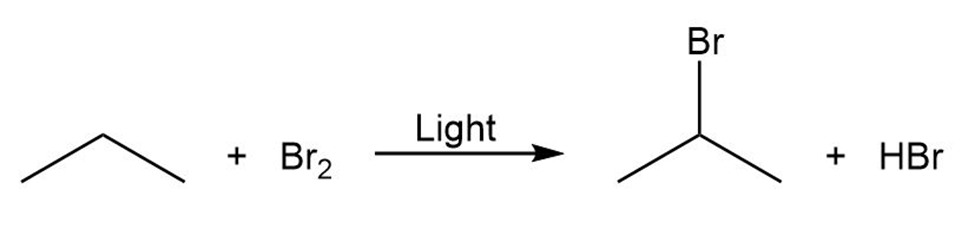
c)
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?
An electrostatic potential map of boron trifluoride is shown. Is likely to be a nucleophile or an electrophile? Draw a Lewis structure for , and explain your answer.
What product would you expect from the reaction of cyclohexene with ? With ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.