Chapter 6: Q20E (page 181)
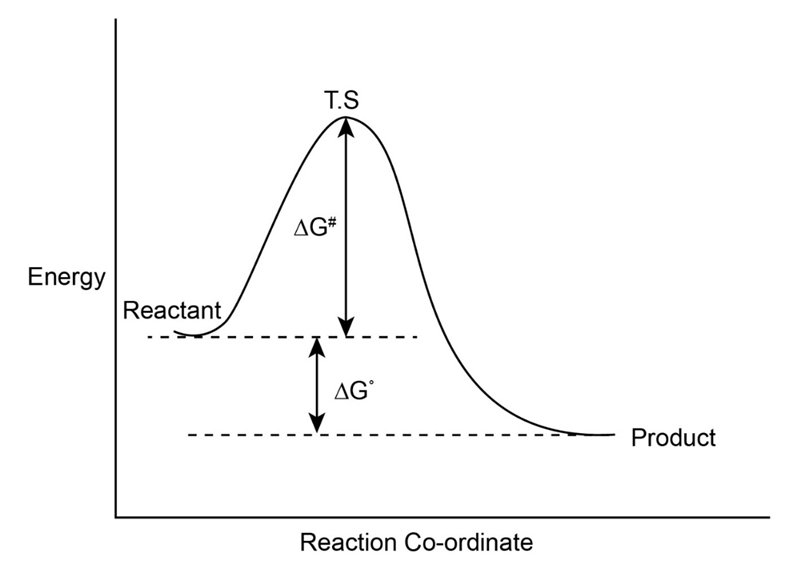
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q20E (page 181)
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the more stable chair conformation of the following molecules, and estimate the amount of strain in each:
Look at the following energy diagram:
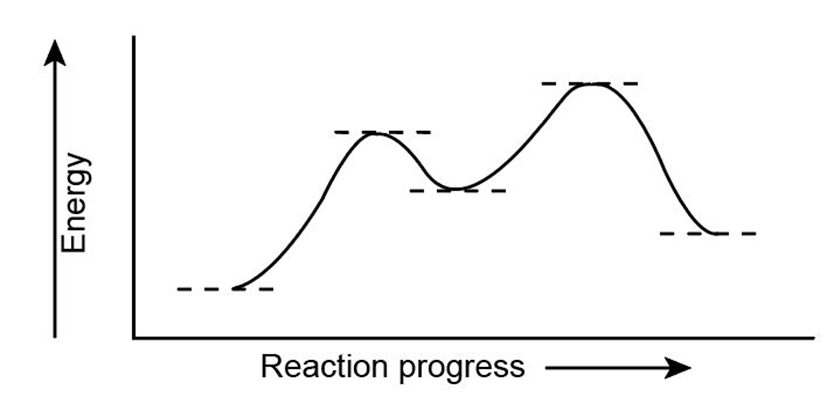
(a) Is for the reaction positive or negative? Label it on the diagram.
(b) How many steps are involved in the reaction?
(c) How many transition states are there? Label them on the diagram.
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step.
Reaction of with 2-methylpropene yields 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.
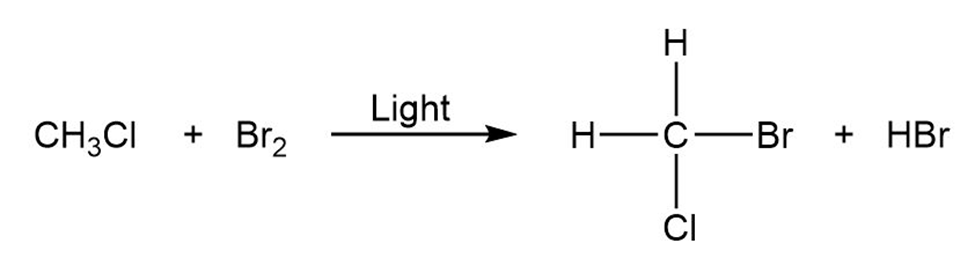
Draw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.
(a)
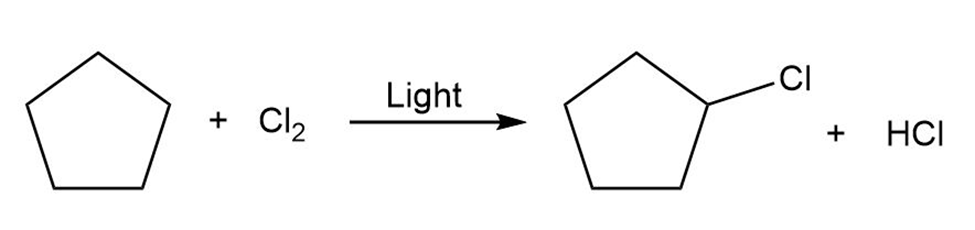
b)

c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.