Chapter 6: Q21E (page 181)
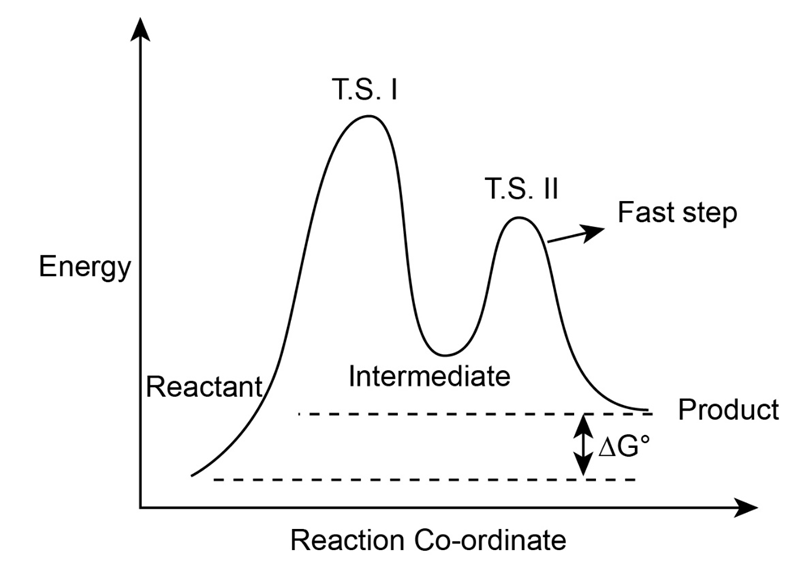
Draw an energy diagram for a two-step reaction with > 1. Label the overall , transition states, and intermediate. Is positive or negative?
Short Answer
Learning Materials
Features
Discover
Chapter 6: Q21E (page 181)
Draw an energy diagram for a two-step reaction with > 1. Label the overall , transition states, and intermediate. Is positive or negative?

All the tools & learning materials you need for study success - in one app.
Get started for free
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement:
1.
2.
3.
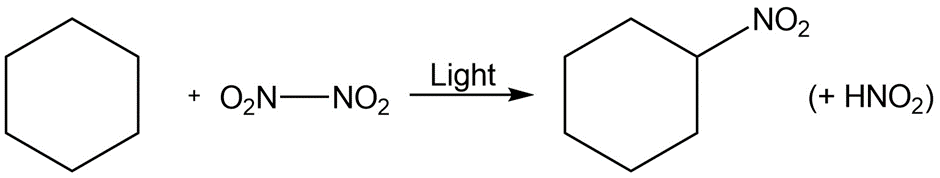
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.

b.
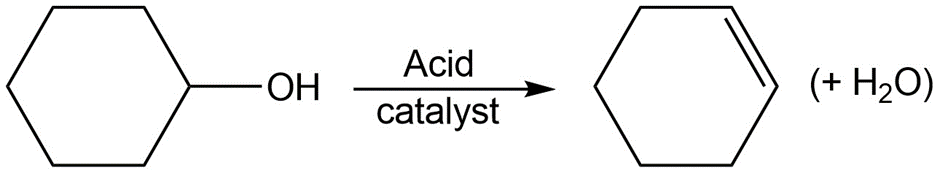
c.
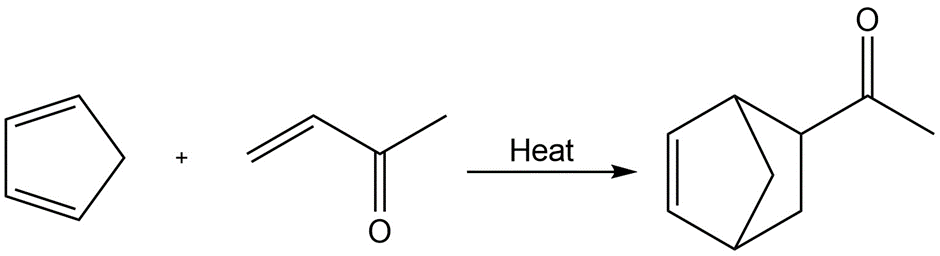
d.
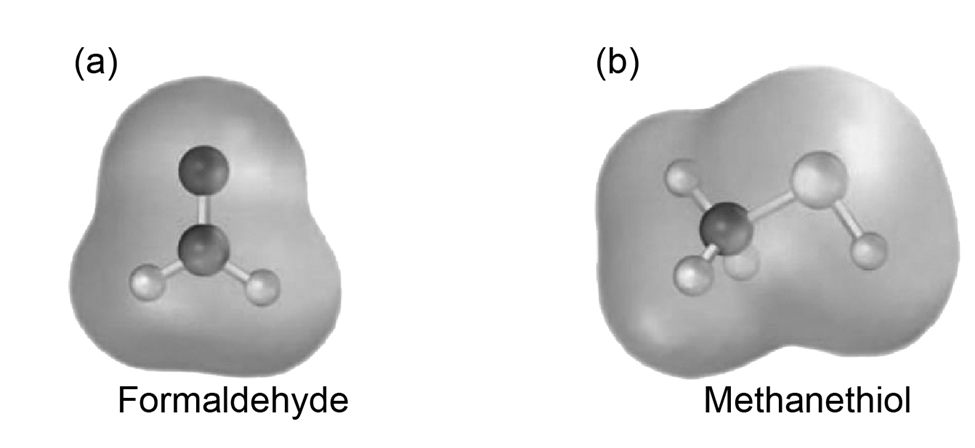
Electrostatic potential maps of (a) formaldehyde and (b) methanethiol are shown. Is the formaldehyde carbon atom likely to be electrophilic or nucleophilic? What about the methanethiol sulfur atom? Explain.
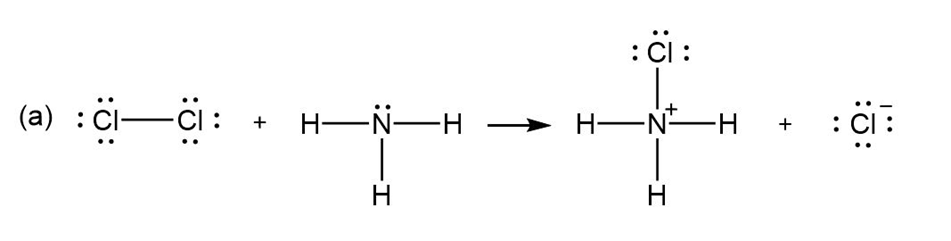
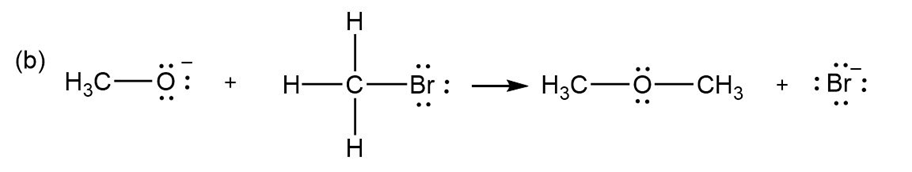
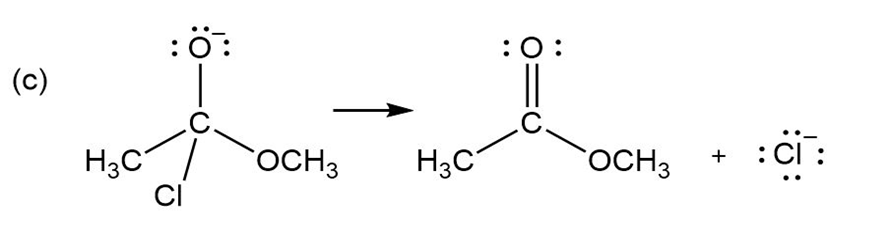
Add curved arrows to the following polar reactions to indicate the flow of electrons in each:
Identify the following reactions as additions, eliminations, substitutions, or rearrangements:
a.

b.
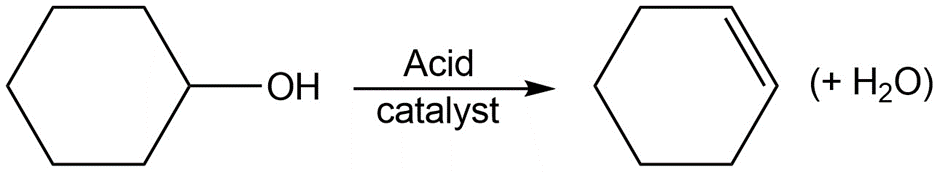
c. 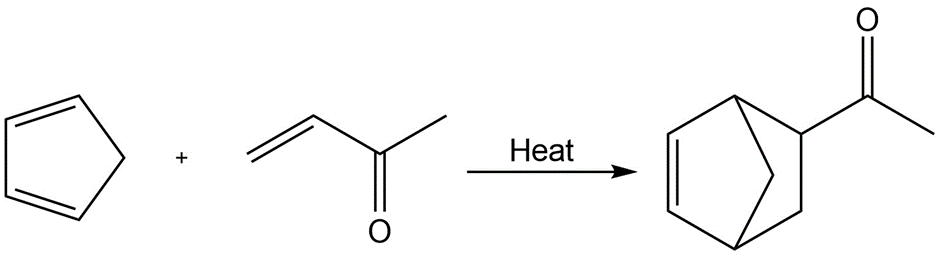
d.
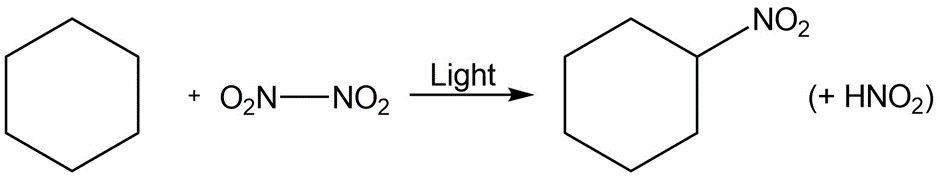
What do you think about this solution?
We value your feedback to improve our textbook solutions.