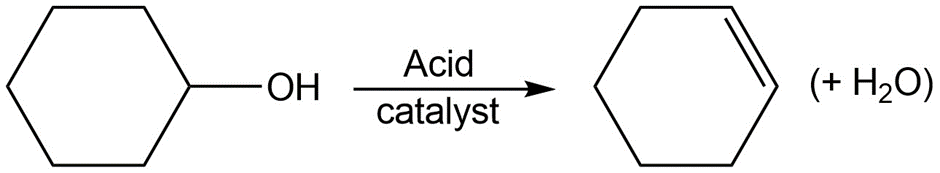
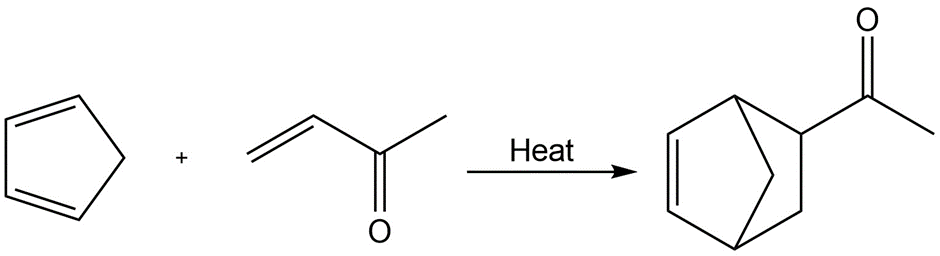
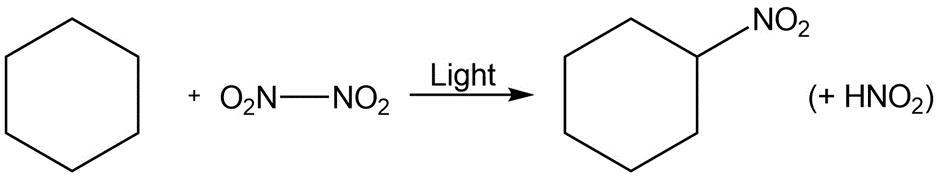
Chapter 6: 42 E (page 181)
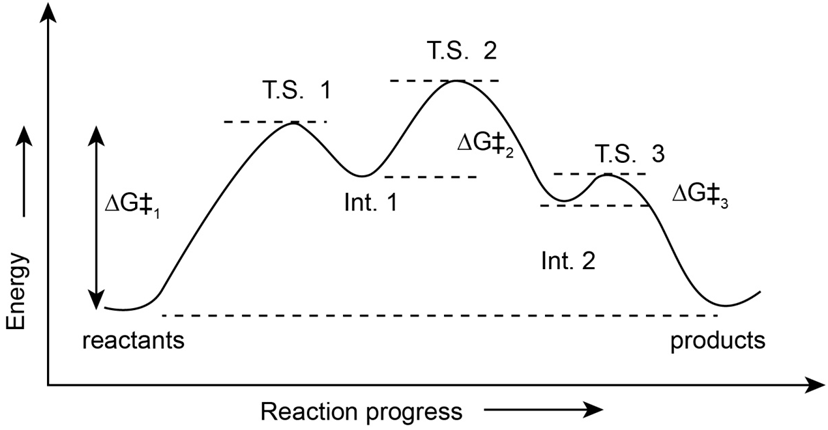
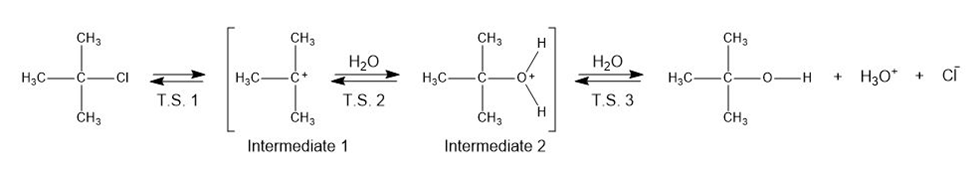
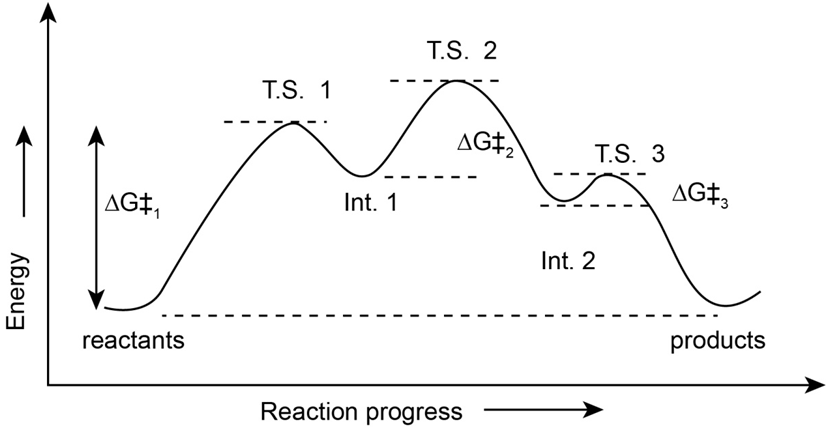
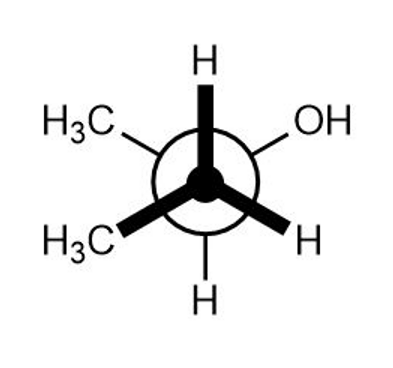
2-Chloro-2-methylpropane reacts with water in three steps to yield 2-methyl-2-propanol. The first step is slower than the second, which in turn is much slower than the third. The reaction takes place slowly at room temperature, and the equilibrium constant is approximately 1.
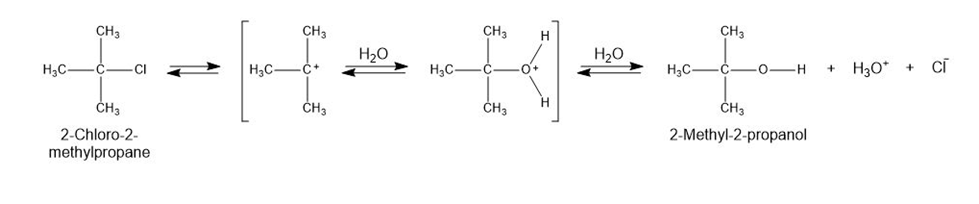
- Give approximate values for and that are consistent with the above information.
- Draw an energy diagram for the reaction, labeling all points of interest and making sure that the relative energy levels on the diagram are consistent with the information given.
Short Answer
a. The value for the Ist step is approximately 80 kJ/mol. The values for the second and third steps are smaller, i.e. 60 kJ/mol for step 2 and 40 kJ/mol for step 3. The value ofis approximately zero.
b. An energy diagram for the reaction is,