Chapter 6: Q14E (page 181)
The following alkyl halide can be prepared by the addition of HBr to two different alkenes. Draw the structures of both (reddish-brown 5 Br).
Short Answer
The structure of alkenes is as follows;
Learning Materials
Features
Discover
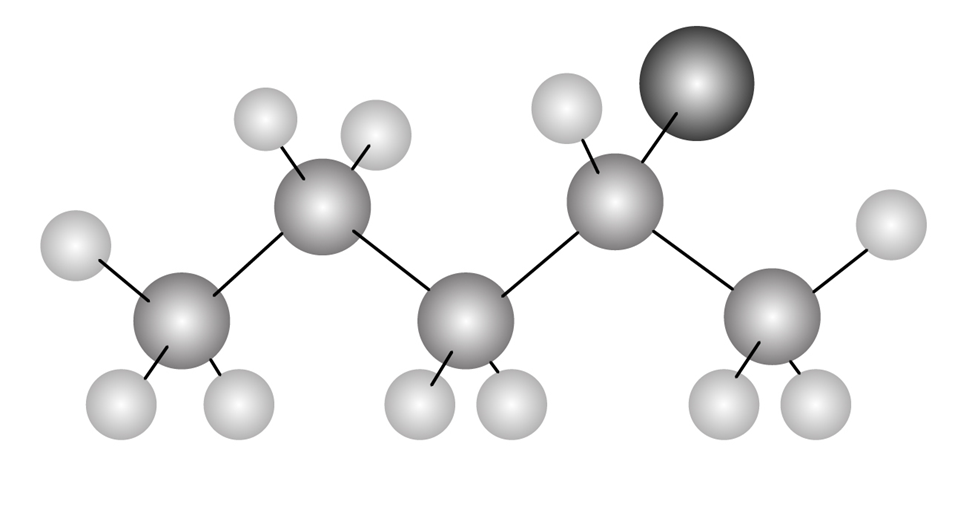
Chapter 6: Q14E (page 181)
The following alkyl halide can be prepared by the addition of HBr to two different alkenes. Draw the structures of both (reddish-brown 5 Br).

The structure of alkenes is as follows;
All the tools & learning materials you need for study success - in one app.
Get started for free
Which reaction is more energetically favored, one with = - 44 or one with = +44 ?
What product would you expect from the reaction of cyclohexene with ? With ?
An electrostatic potential map of boron trifluoride is shown. Is likely to be a nucleophile or an electrophile? Draw a Lewis structure for , and explain your answer.
Which reaction is likely to be more exergonic, one with =1000 or one with = 0.001?
The following structure represents the carbocation intermediate formed in the addition reaction of H-Br to two different alkenes. Draw the structures of both.
What do you think about this solution?
We value your feedback to improve our textbook solutions.