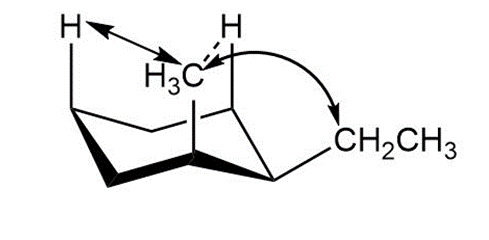
Chapter 6: 18-B (page 149)
Draw the more stable chair conformation of the following molecules, and estimate the amount of strain in each:
Short Answer
The more stable chair conformation is given below and 11.4 kJ/mol strain present.
Learning Materials
Features
Discover
Chapter 6: 18-B (page 149)
Draw the more stable chair conformation of the following molecules, and estimate the amount of strain in each:
The more stable chair conformation is given below and 11.4 kJ/mol strain present.

All the tools & learning materials you need for study success - in one app.
Get started for free
The following structure represents the carbocation intermediate formed in the addition reaction of H-Br to two different alkenes. Draw the structures of both.
What product would you expect from the reaction of cyclohexene with ? With ?
Classify each of the following reactions as an addition, elimination, substitution, or rearrangement:
1.
2.
3.
Look at the following energy diagram:

(a) Is for the reaction positive or negative? Label it on the diagram.
(b) How many steps are involved in the reaction?
(c) How many transition states are there? Label them on the diagram.
2-Chloro-2-methylpropane reacts with water in three steps to yield 2-methyl-2-propanol. The first step is slower than the second, which in turn is much slower than the third. The reaction takes place slowly at room temperature, and the equilibrium constant is approximately 1.

What do you think about this solution?
We value your feedback to improve our textbook solutions.