Chapter 6: 6-35c (page 181)
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:
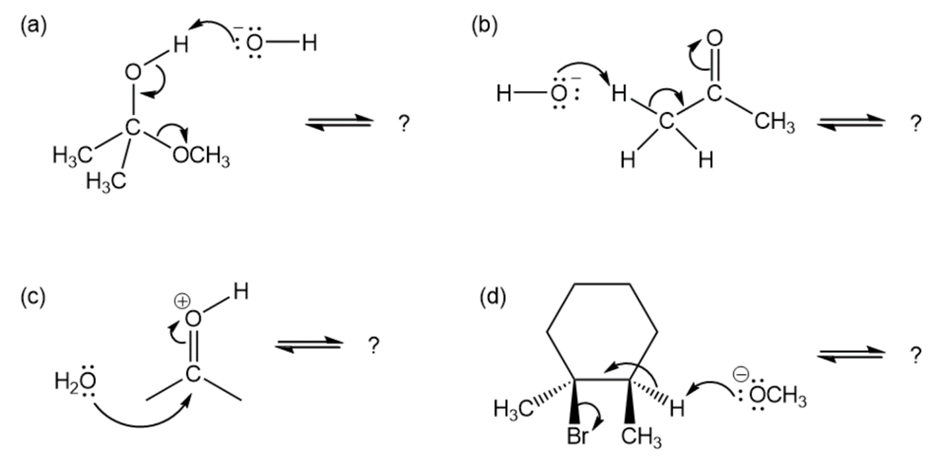
Short Answer
(c)
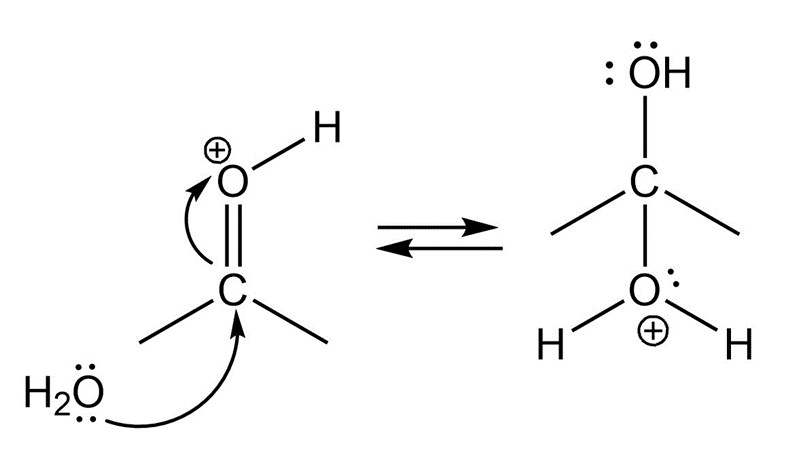
Formation of intermediate
Learning Materials
Features
Discover
Chapter 6: 6-35c (page 181)
Follow the flow of electrons indicated by the curved arrows in each of the following polar reactions, and predict the products that result:

(c)

Formation of intermediate
All the tools & learning materials you need for study success - in one app.
Get started for free
Which reaction is likely to be more exergonic, one with =1000 or one with = 0.001?
Reaction of with 2-methylpropene yields 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.
Draw an energy diagram for a one-step reaction with . Label the parts of the diagram corresponding to reactants, products, transition state, , and . Is positive or negative?
The addition of water to ethylene to yield ethanol has the following thermodynamic parameters:
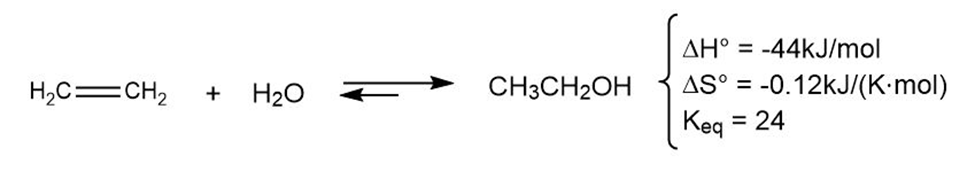
(a) Is the reaction exothermic or endothermic?
(b) Is the reaction favorable (spontaneous) or unfavorable (nonspontaneous) at room temperature (298 K)?
What product would you expect from the reaction of cyclohexene with ? With ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.