Chapter 19: Q83E (page 648)
Propose structures for ketones or aldehydes that have the following 1H NMR spectra:
(a) C9H10O2
IR: 1695 cm-1
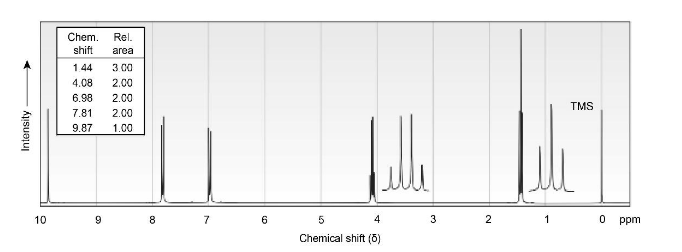
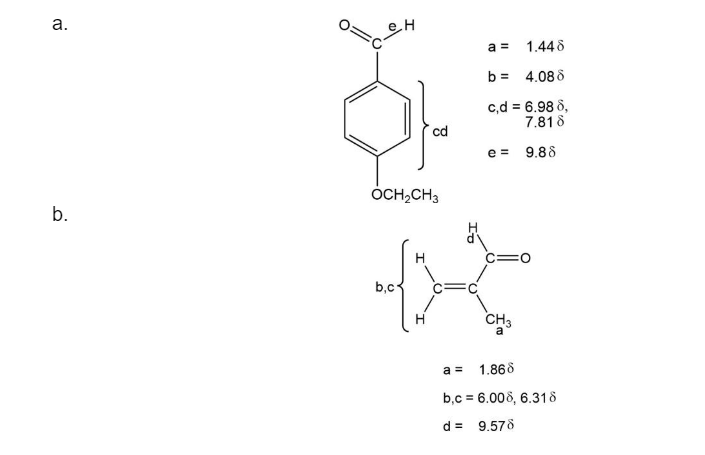
(b) C4H6O
IR: 1690 cm-1

Short Answer
The structure of the compound is:
Learning Materials
Features
Discover
Chapter 19: Q83E (page 648)
Propose structures for ketones or aldehydes that have the following 1H NMR spectra:
(a) C9H10O2
IR: 1695 cm-1

(b) C4H6O
IR: 1690 cm-1

The structure of the compound is:

All the tools & learning materials you need for study success - in one app.
Get started for free
Cyclohexanone forms a cyanohydrin in good yield but 2,2,6-trimethylcyclohexanonedoes not. Explain.
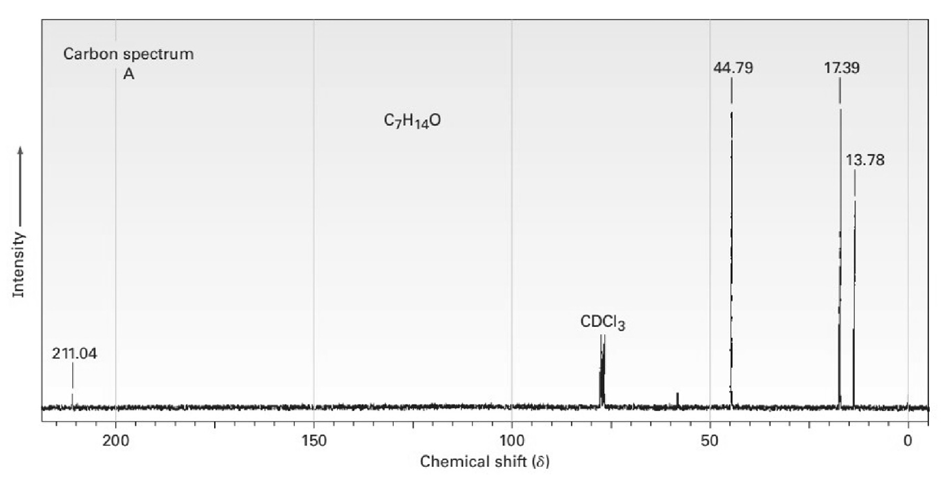
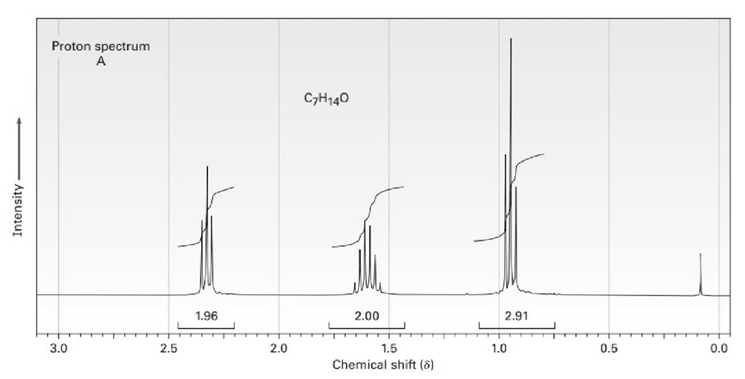
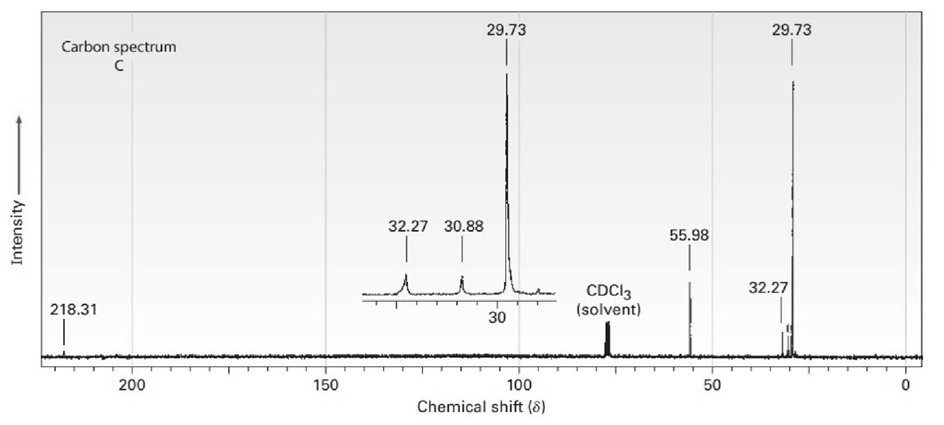
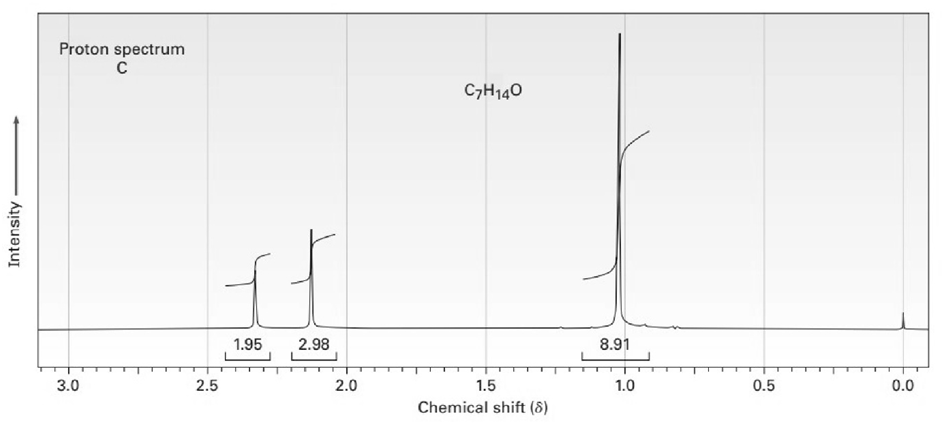
The proton and carbon NMR spectra for each of three isomeric ketones with the formula C7H14Oare shown below. Assign a structure to each pair of spectra.



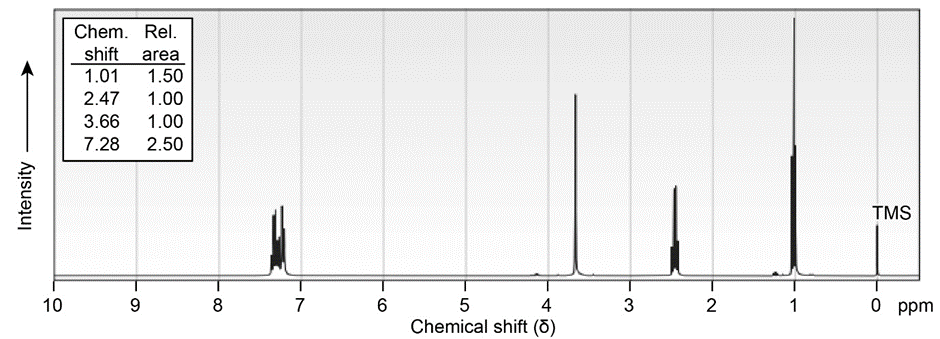
Propose structures for ketones or aldehydes that have the following \(^1H\;NMR\) spectra:
(a) \({C_{10}}{H_{12}}O\)
IR: 1710 \(c{m^{ - 1}}\)
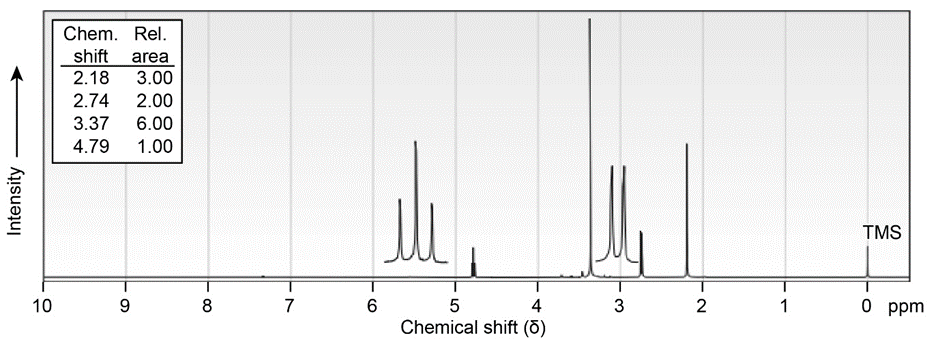
(b)\({C_6}{H_{12}}{O_3}\)
IR: 1715\(c{m^{ - 1}}\)
Reaction of 2-butanone with HCN yields a chiral product. What stereochemistry does the product have? Is it optically active?
Show the products you would obtain by acid-catalyzed reaction of cyclohexanonewith ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2)2NH.
What do you think about this solution?
We value your feedback to improve our textbook solutions.