Chapter 19: Q19-10P (page 624)
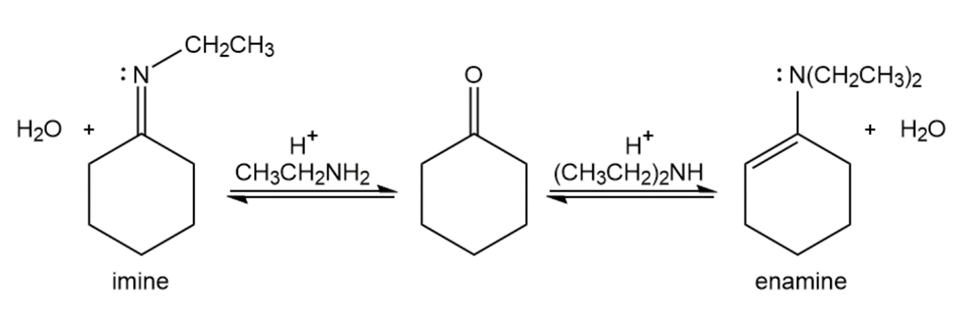
Show the products you would obtain by acid-catalyzed reaction of cyclohexanonewith ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2)2NH.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q19-10P (page 624)
Show the products you would obtain by acid-catalyzed reaction of cyclohexanonewith ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2)2NH.

All the tools & learning materials you need for study success - in one app.
Get started for free
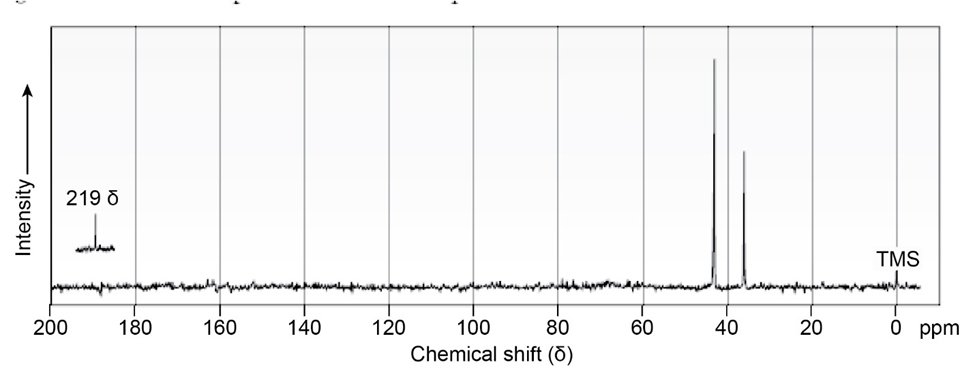
Compound A, , has an intense IR absorption at 1750 and gives the 13C NMR spectrum shown. Propose a structure for A.
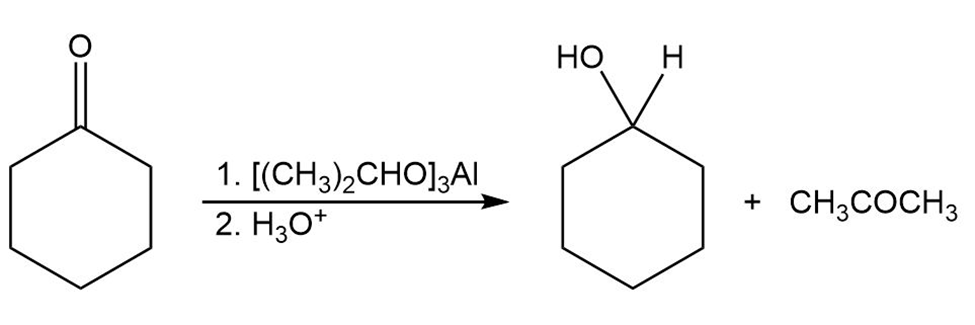
The Meerwein–Ponndorf–Verley reaction involves the reduction of a ketone by treatment with an excess of aluminum triisopropoxide,.The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.
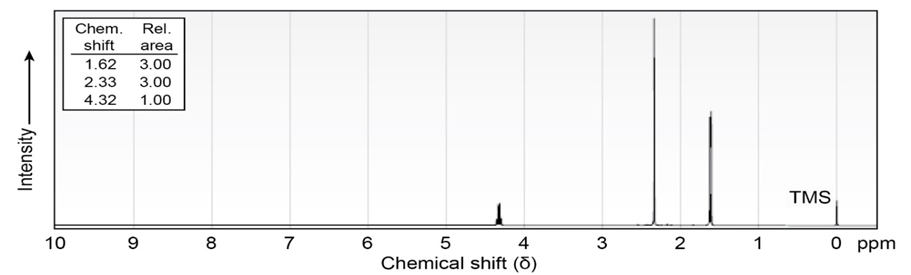
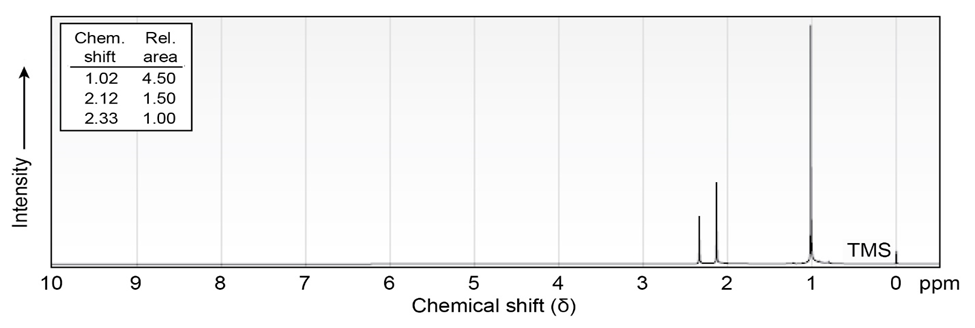
Propose structures for ketones or aldehydes that have the following 1H
NMR spectra:
(a)
IR: 1715
(b)
IR: 1710
Draw structures of compounds that fit the following descriptions:
Draw structures corresponding to the following names:
What do you think about this solution?
We value your feedback to improve our textbook solutions.