Chapter 19: Q71E (page 648)
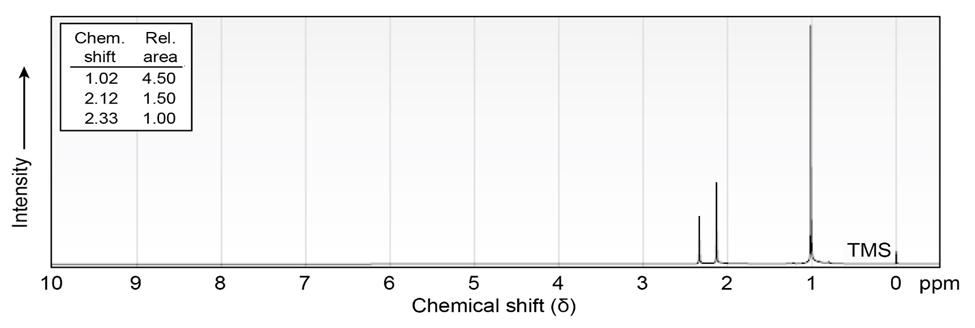
Propose structures for ketones or aldehydes that have the following 1H
NMR spectra:
(a)
IR: 1715

(b)
IR: 1710
Short Answer
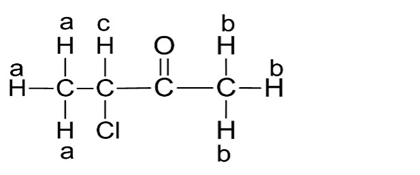
The structure of the given compound is:
a.
b.
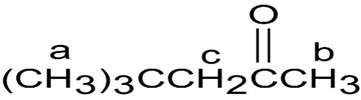
Learning Materials
Features
Discover
Chapter 19: Q71E (page 648)
Propose structures for ketones or aldehydes that have the following 1H
NMR spectra:
(a)
IR: 1715

(b)
IR: 1710

The structure of the given compound is:
a.

b.
All the tools & learning materials you need for study success - in one app.
Get started for free
The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotopeis also available. When an aldehyde or ketone is dissolvedin-enriched water, the isotopic label becomes incorporated into thecarbonyl group. Explain.
where O =
Acid-catalyzed dehydration of 3-hydroxy-3-phenylcyclohexanone leads to an unsaturated ketone. What possible structures are there for the product? At what position in the IR spectrum would you expect each to absorb? If the actual product has an absorption at , what is its structure?
At what position would you expect to observe IR absorptions for the following molecules?
a.
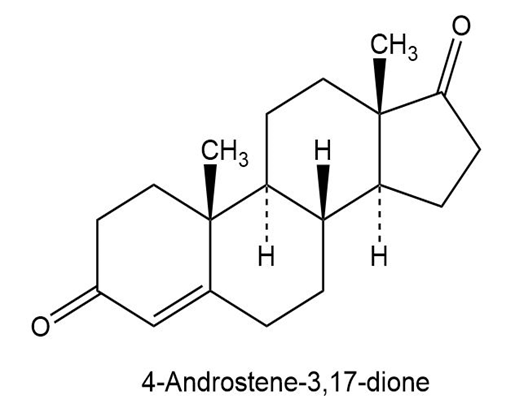
b.
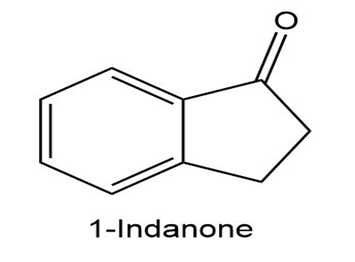
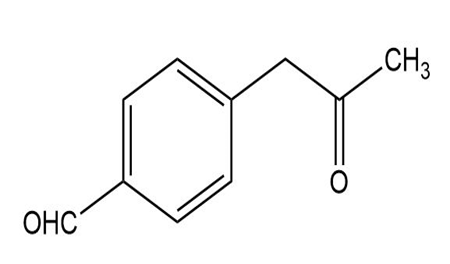
c.

d.
Treatment of 2-cyclohexenone with HCN/KCN yields a saturated keto nitrile rather than an unsaturated cyanohydrin. Show the structure of the product,
and propose a mechanism for the reaction.
The following molecular model represents a tetrahedral intermediateresulting from addition of a nucleophile to an aldehyde or ketone. Identifythe reactants, and write the structure of the final product when thenucleophilic addition reaction is complete.
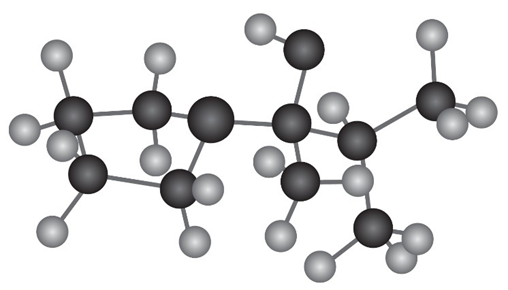
What do you think about this solution?
We value your feedback to improve our textbook solutions.