Chapter 19: Q18P (page 635)
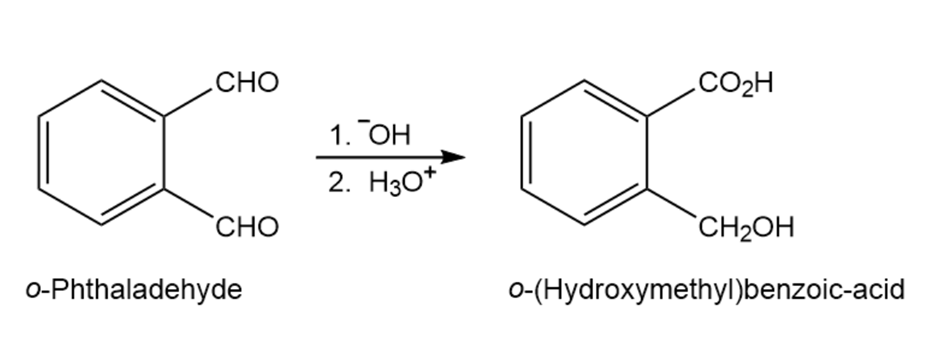
When o-phthalaldehyde is treated with base, o-(hydroxymethyl)benzoic acid is formed. Show the mechanism of this reaction.
Short Answer
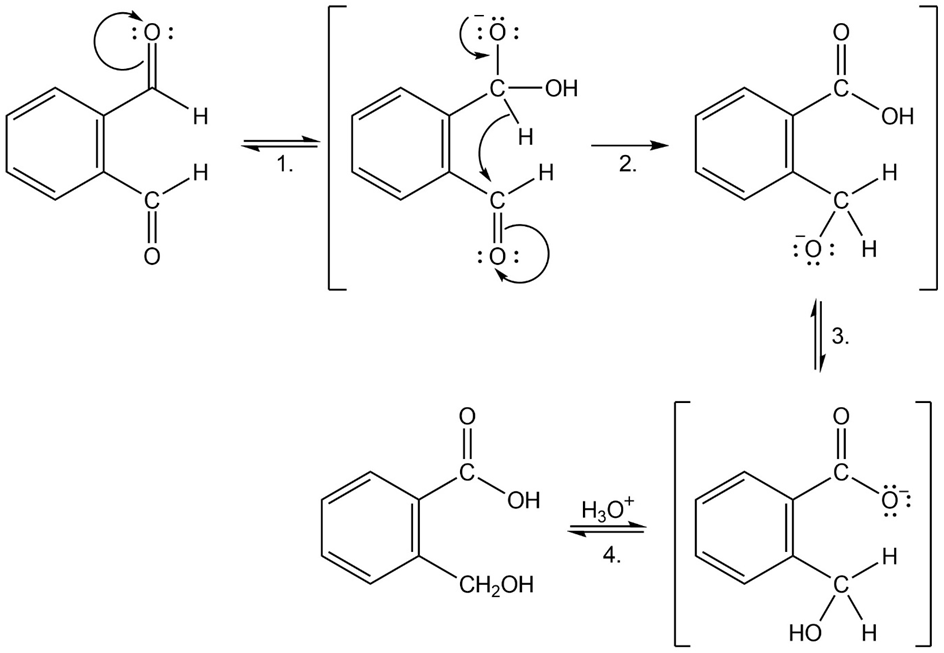
Formation of desired product
Learning Materials
Features
Discover
Chapter 19: Q18P (page 635)
When o-phthalaldehyde is treated with base, o-(hydroxymethyl)benzoic acid is formed. Show the mechanism of this reaction.


Formation of desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
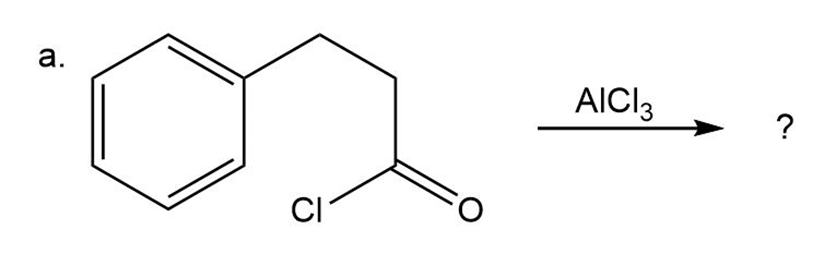
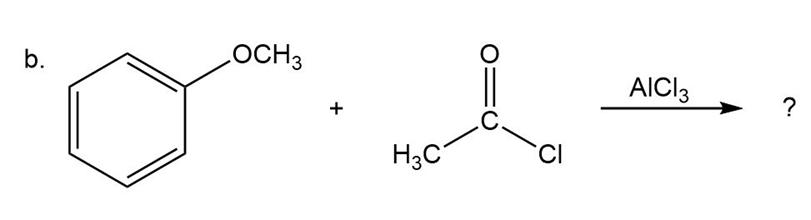
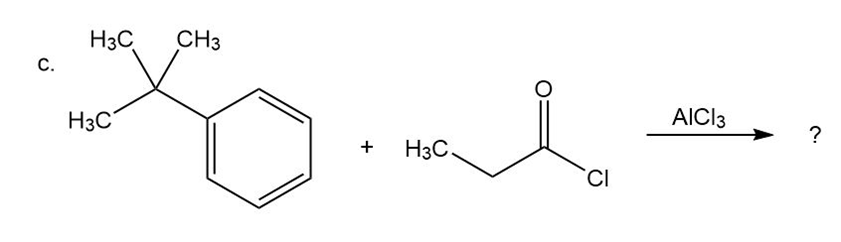
Predict the product(s) and provide the mechanism for each reactionbelow. What does each mechanism have in common?
Draw structures corresponding to the following names:
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
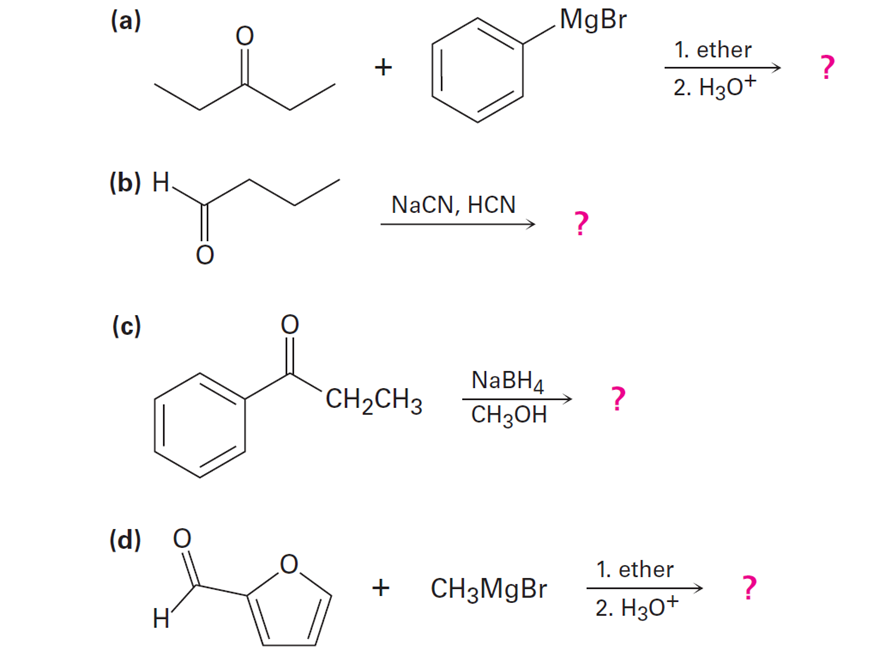
How might conjugate addition reactions of lithium diorganocopper reagents be used to synthesize the following compounds?
Cyclohexanone forms a cyanohydrin in good yield but 2,2,6-trimethylcyclohexanonedoes not. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.