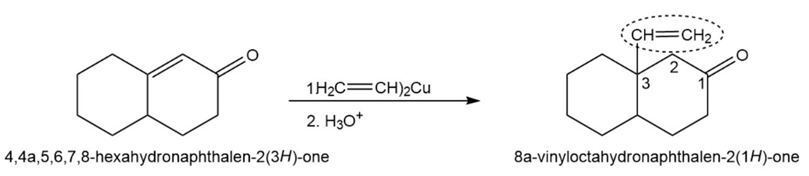
Chapter 19: Q22P (page 624)
How might conjugate addition reactions of lithium diorganocopper reagents be used to synthesize the following compounds?
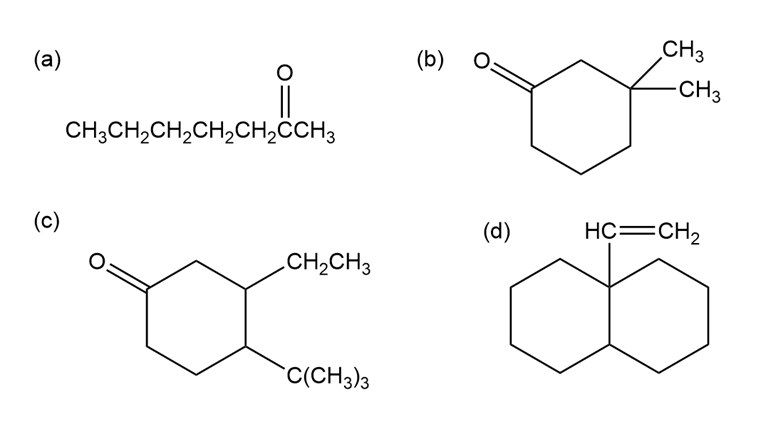
Short Answer
(a)
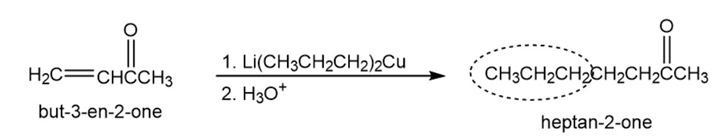
(b)
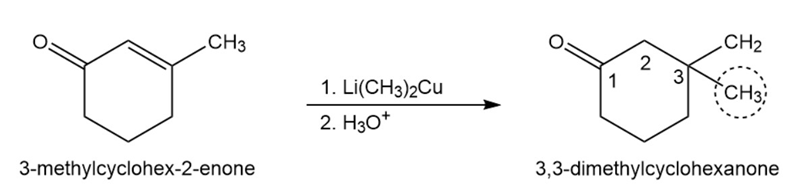
(c)
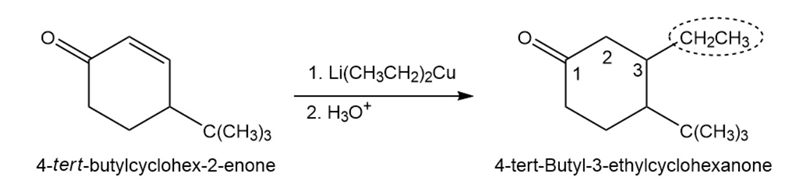
(d)
Learning Materials
Features
Discover
Chapter 19: Q22P (page 624)
How might conjugate addition reactions of lithium diorganocopper reagents be used to synthesize the following compounds?

(a)
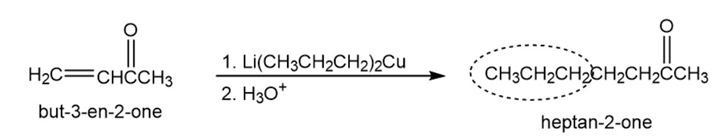
(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw and name the seven aldehydes and ketones with the formula . Which are chiral?
Question: Predict the products of the wolff-Kishner reduction reactions below. Provide the electron pushing mechanism for each, beginning from the hydrazone intermediate.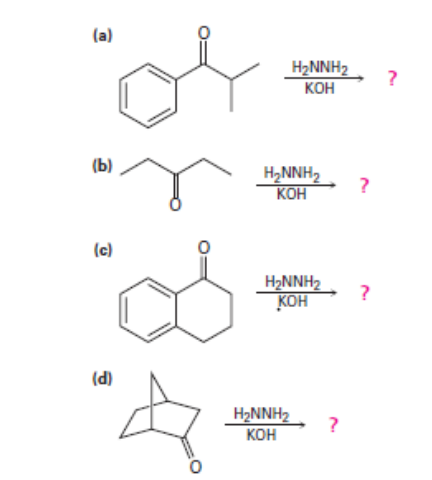
Compound A, M+= 86, shows an IR absorption at 1730 cm-and a very simple 1HNMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.
Cyclohexanone forms a cyanohydrin in good yield but 2,2,6-trimethylcyclohexanonedoes not. Explain.
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)
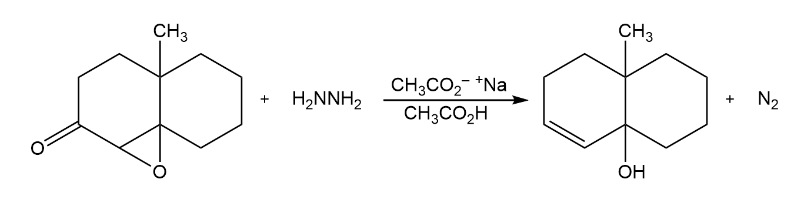
What do you think about this solution?
We value your feedback to improve our textbook solutions.