Chapter 19: Q53E (page 648)
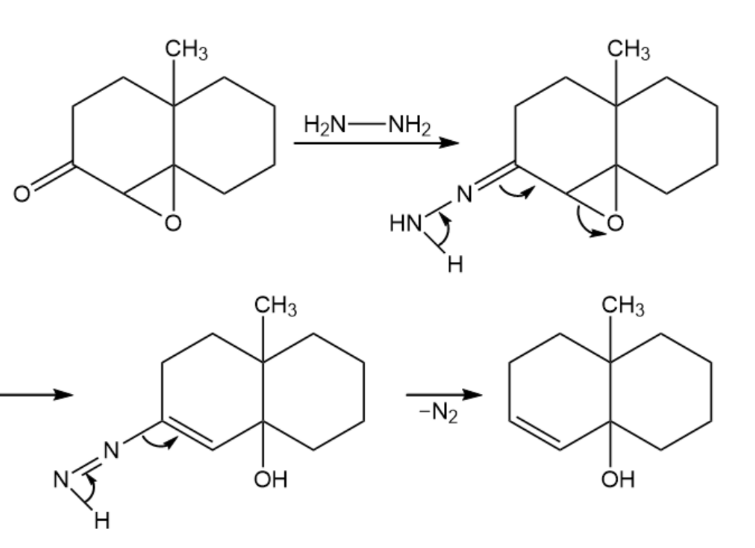
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)

Short Answer
Learning Materials
Features
Discover
Chapter 19: Q53E (page 648)
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)


All the tools & learning materials you need for study success - in one app.
Get started for free
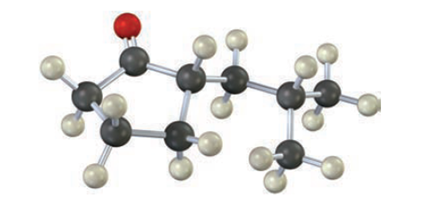
Describe the prominent IR absorptions and mass spectral peaks you would expect for the following compound:
The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotopeis also available. When an aldehyde or ketone is dissolvedin-enriched water, the isotopic label becomes incorporated into thecarbonyl group. Explain.
where O =
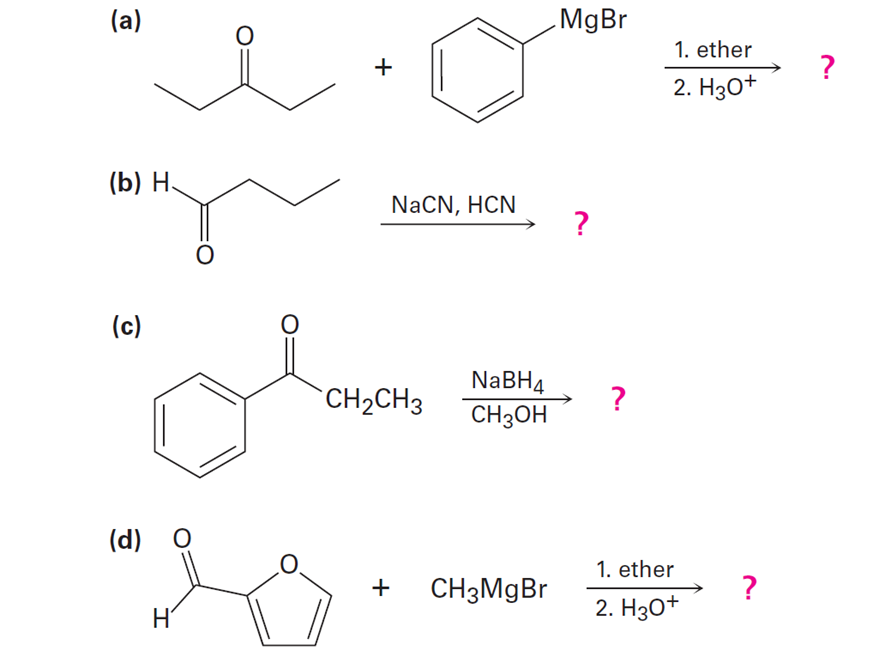
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
How would you synthesize the following compounds from cyclohexanone?
Draw and name the seven aldehydes and ketones with the formula . Which are chiral?
What do you think about this solution?
We value your feedback to improve our textbook solutions.