Chapter 19: Q52E (page 648)
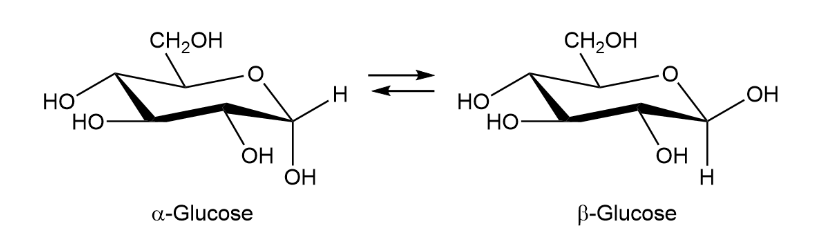
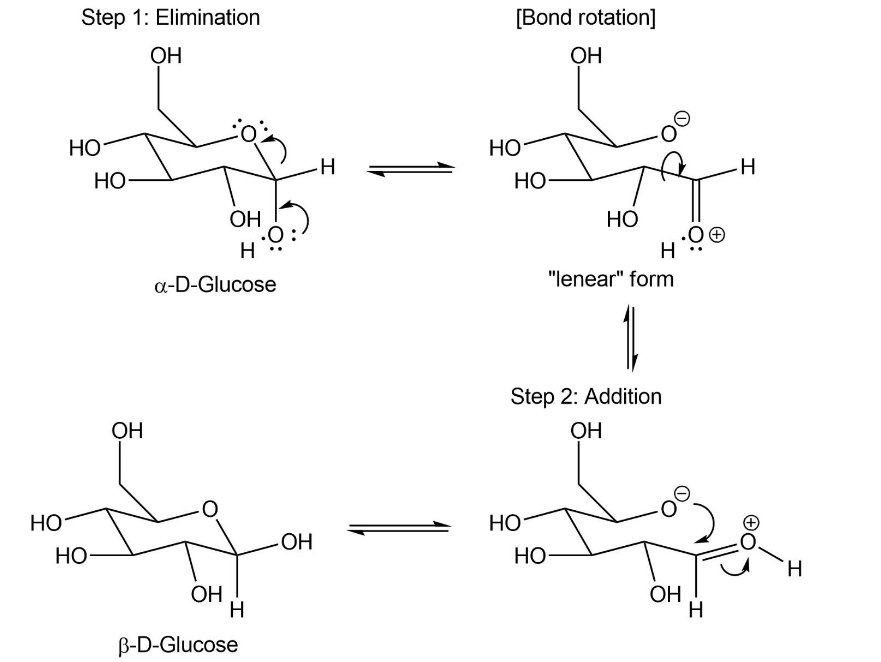
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q52E (page 648)
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.


All the tools & learning materials you need for study success - in one app.
Get started for free
The enamine prepared from acetone and dimethylamine is shown in its
lowest-energy form.
(a) What is the geometry and hybridization of the nitrogen atom?
(b) What orbital on nitrogen holds the lone pair of electrons?
(c) What is the geometric relationship between the p orbitals of the
double bond and the nitrogen orbital that holds the lone pair? Why
do you think this geometry represents the minimum energy?
Choose the structure that best fits the IR spectrum shown.
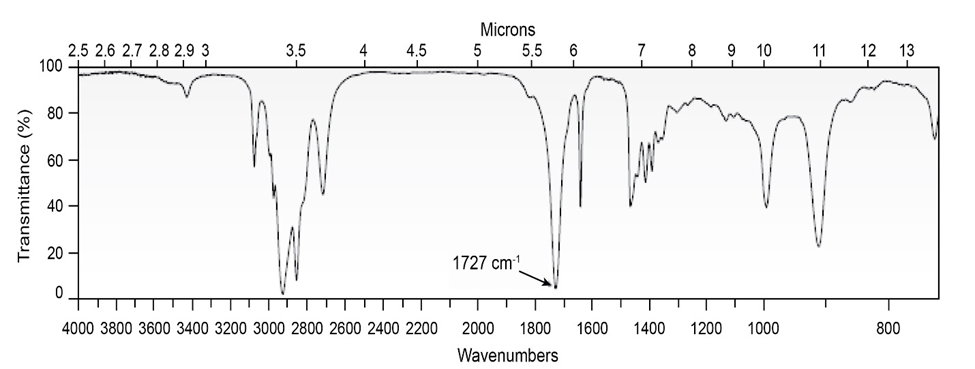
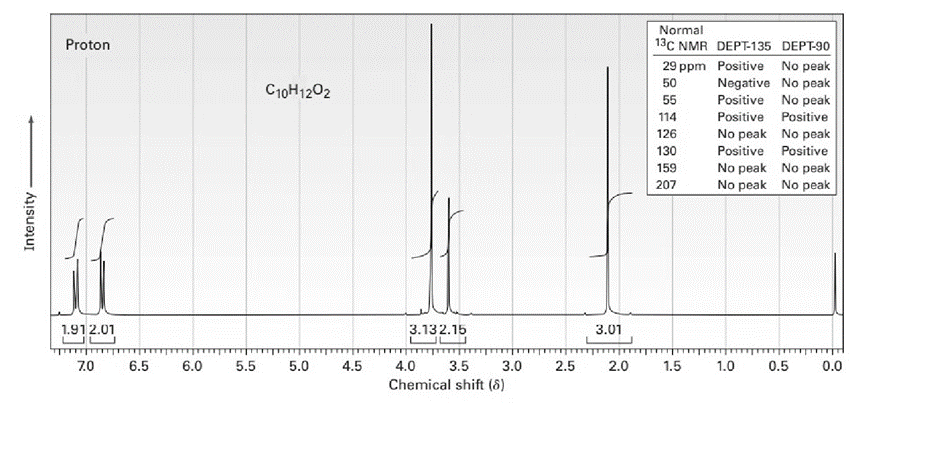
The proton NMR spectrum for a compound with formulais shown below. The infrared spectrum has a strong band at. The broadband-decoupledNMR spectral results are tabulated along with the DEPT-135 and DEPT-90 information. Draw the structure of this compound.
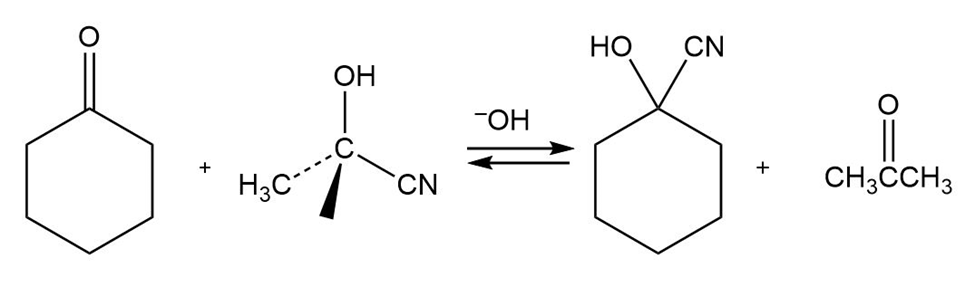
When cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, cyclohexanone cyanohydrin and acetone are formed. Propose a mechanism.
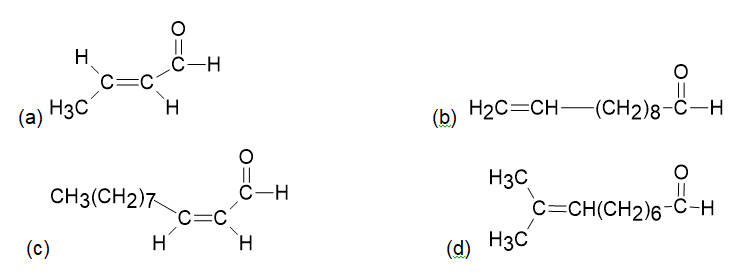
At what position would you expect to observe IR absorptions for the following molecules?
a.
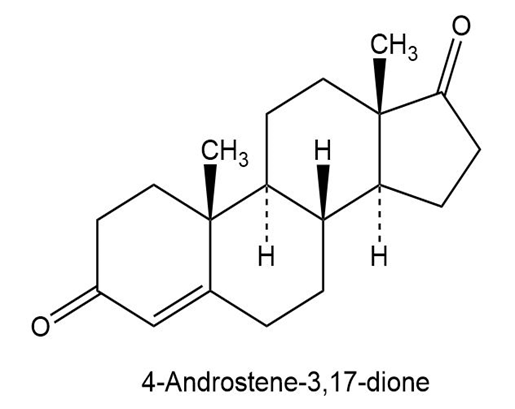
b.
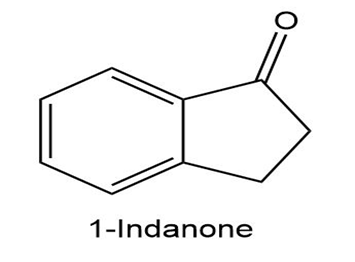
c.
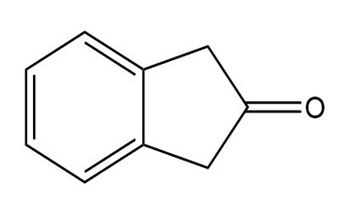
d.
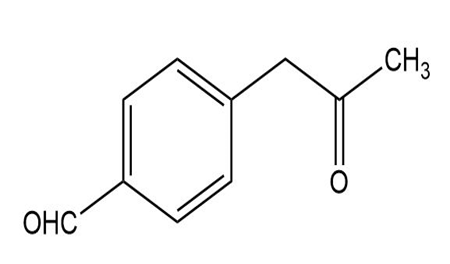
What do you think about this solution?
We value your feedback to improve our textbook solutions.