Chapter 19: Q51E (page 648)
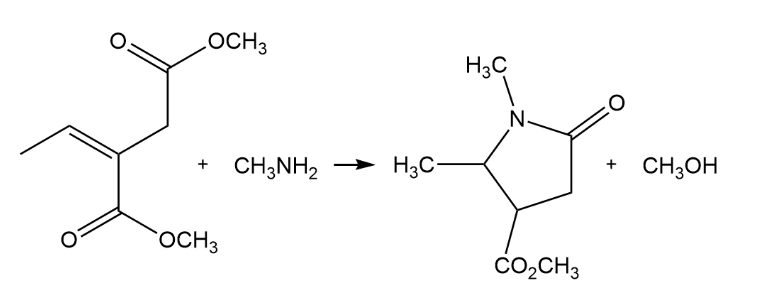
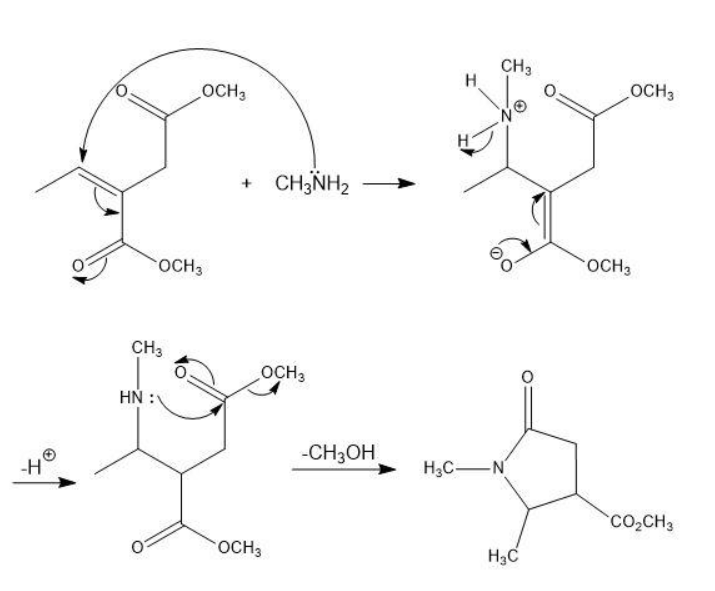
Primary amines react with esters to yield amides: RCO2R' + R"NH2 RCONHR" + R'OH . Propose a mechanism for the following reaction of an a b-unsaturated ester.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q51E (page 648)
Primary amines react with esters to yield amides: RCO2R' + R"NH2 RCONHR" + R'OH . Propose a mechanism for the following reaction of an a b-unsaturated ester.


All the tools & learning materials you need for study success - in one app.
Get started for free
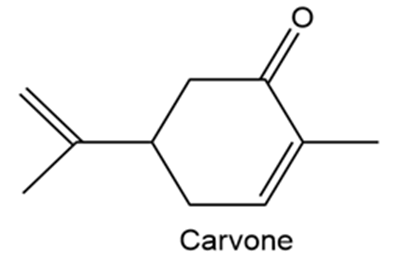
Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents?
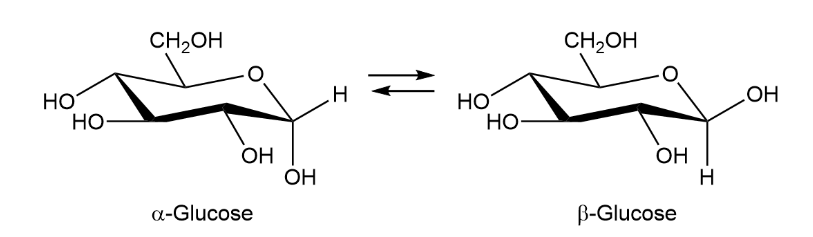
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.
When dissolved in water, trichloroacetaldehyde exists primarily as its hydrate, called chloral hydrate. Show the structure of chloral hydrate.
How might conjugate addition reactions of lithium diorganocopper reagents be used to synthesize the following compounds?
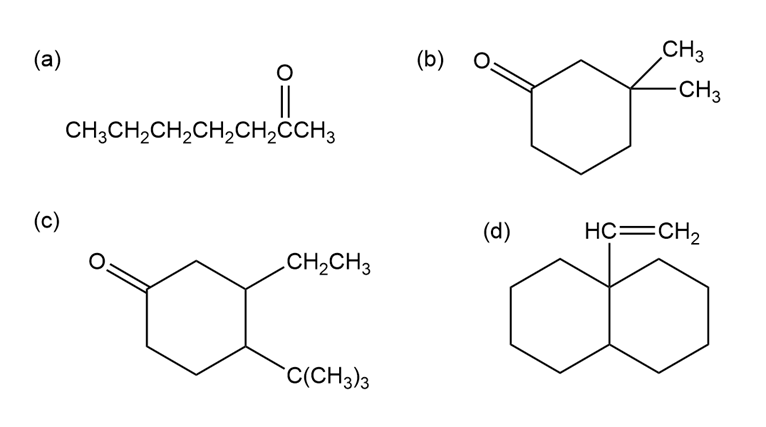
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds?
What do you think about this solution?
We value your feedback to improve our textbook solutions.