Chapter 19: Q19-7P (page 616)
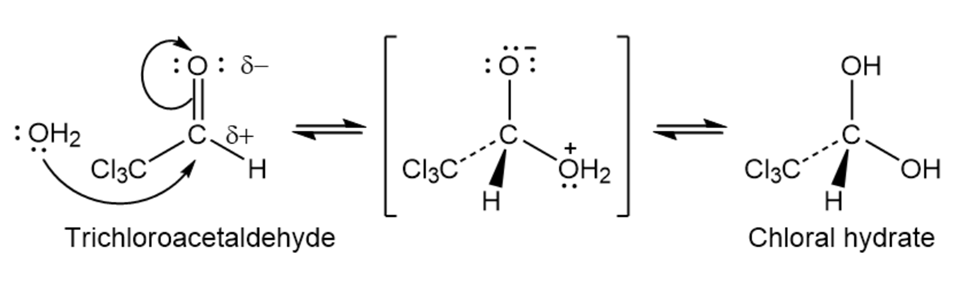
When dissolved in water, trichloroacetaldehyde exists primarily as its hydrate, called chloral hydrate. Show the structure of chloral hydrate.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q19-7P (page 616)
When dissolved in water, trichloroacetaldehyde exists primarily as its hydrate, called chloral hydrate. Show the structure of chloral hydrate.

All the tools & learning materials you need for study success - in one app.
Get started for free
The SN2 reaction of (dibromomethyl)benzene, C6H5CHBr2, withNaOH yields benzaldehyde rather than (dihydroxymethyl)benzene,C6H5CH(OH)2. Explain.
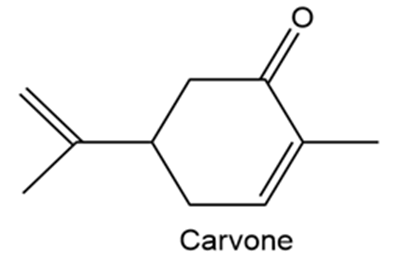
Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents?
Compound A, M+= 86, shows an IR absorption at 1730 cm-and a very simple 1HNMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.
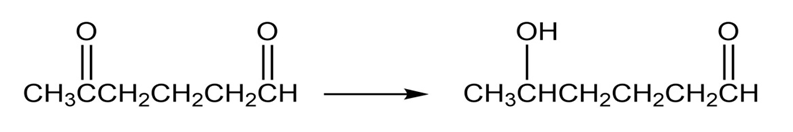
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19-4 that aldehydes are more reactive than ketones towards nucleophilic addition.)
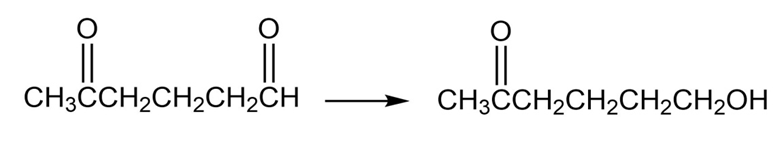
b.
Propose structures for ketones or aldehydes that have the following 1H
NMR spectra:
(a)
IR: 1715
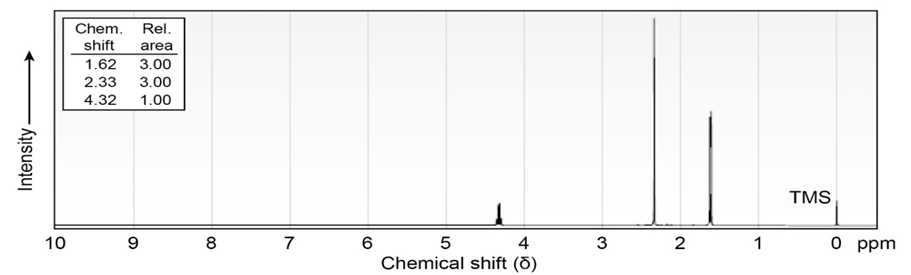
(b)
IR: 1710

What do you think about this solution?
We value your feedback to improve our textbook solutions.