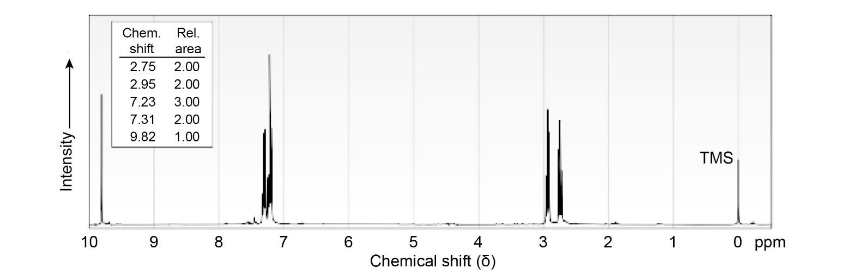
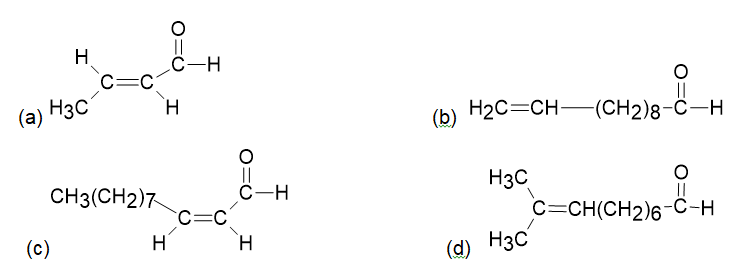
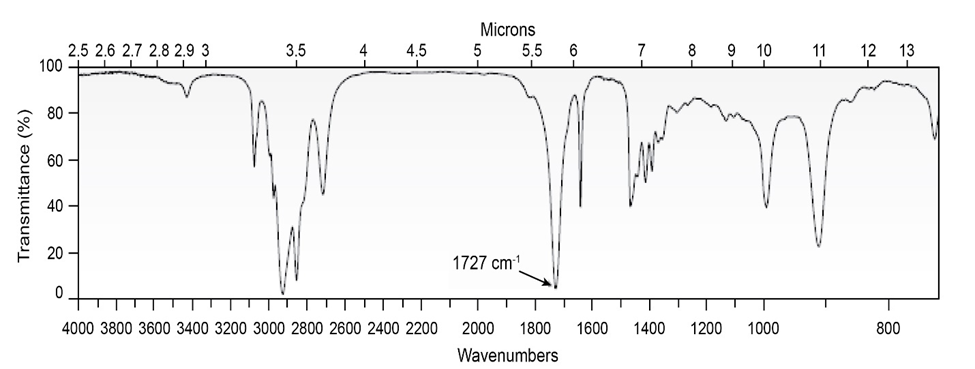
Chapter 19: Q19-5P (page 613)
Treatment of an aldehyde or ketone with cyanide ion (), followed by protonation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrin.
Show the structure of the cyanohydrin obtained from cyclohexanone.
Short Answer

 Structure of cyanohydrin formed
Structure of cyanohydrin formed