Chapter 19: Q79E (page 648)
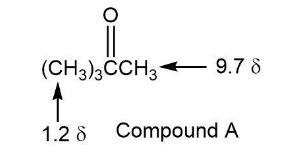
Compound A, M+= 86, shows an IR absorption at 1730 cm-and a very simple 1HNMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.
Short Answer
The structure of compound a is:
Learning Materials
Features
Discover
Chapter 19: Q79E (page 648)
Compound A, M+= 86, shows an IR absorption at 1730 cm-and a very simple 1HNMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.
The structure of compound a is:

All the tools & learning materials you need for study success - in one app.
Get started for free
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds?
Question: Predict the products of the wolff-Kishner reduction reactions below. Provide the electron pushing mechanism for each, beginning from the hydrazone intermediate.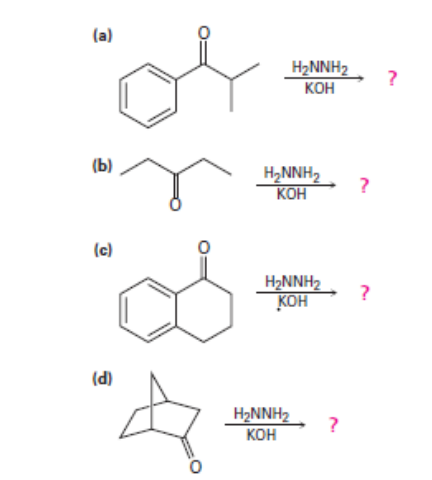
p-Nitrobenzaldehyde is more reactive toward nucleophilic additions than p-methoxybenzaldehyde. Explain.
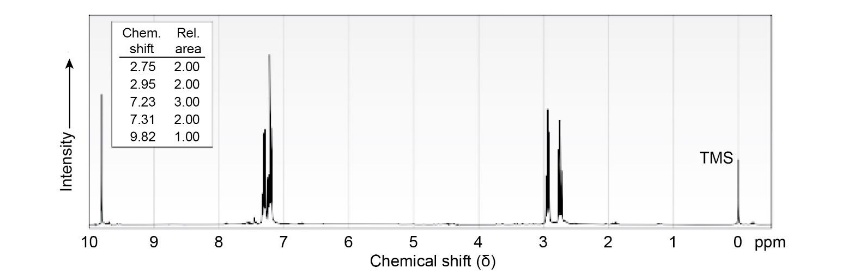
The 1HNMR spectrum shown is that of a compound isomeric with the one in Problem 19-81. This isomer has an IR absorption at 1730 cm-1. Propose a structure.
[Note: Aldehyde protons (CHO) often show low coupling constants to adjacent hydrogens, so the splitting of aldehyde signals is not always apparent.]
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)
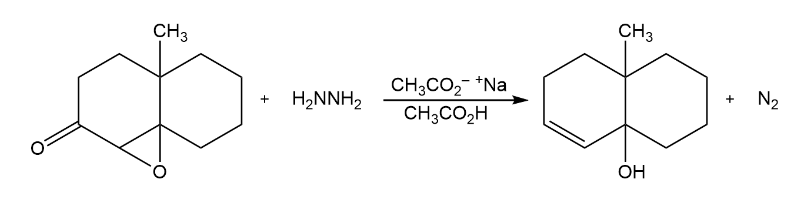
What do you think about this solution?
We value your feedback to improve our textbook solutions.