Chapter 19: Q60E (page 648)
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds?
- 2-Pentanol
- 1-Butanol
- 1-Phenylcyclohexanol
- Diphenylmethanol
Short Answer
a)
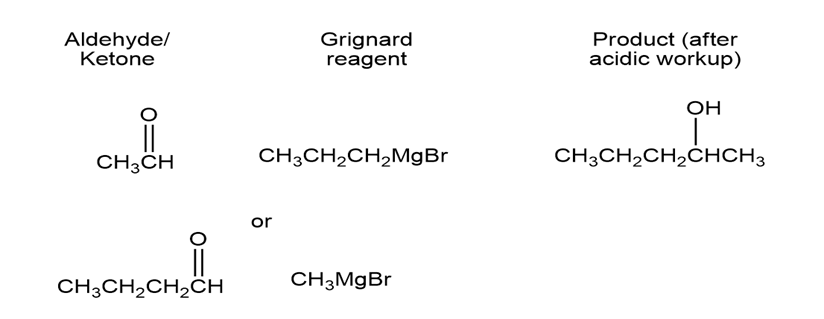
b)
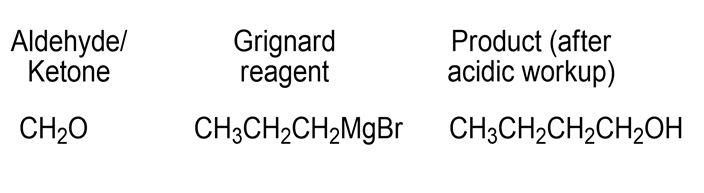
c)

d)

Learning Materials
Features
Discover
Chapter 19: Q60E (page 648)
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds?
a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw and name the seven aldehydes and ketones with the formula . Which are chiral?
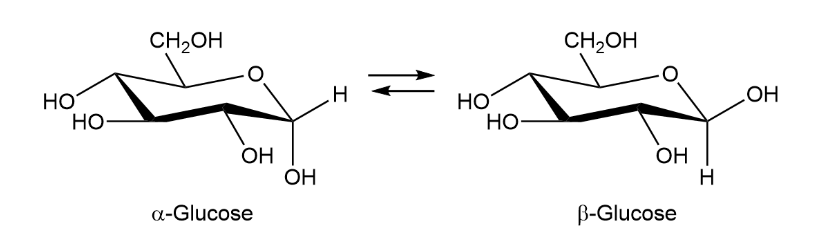
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.
What carbonyl compound and what phosphorus ylide might you use to prepare each of the following compounds?
(a)
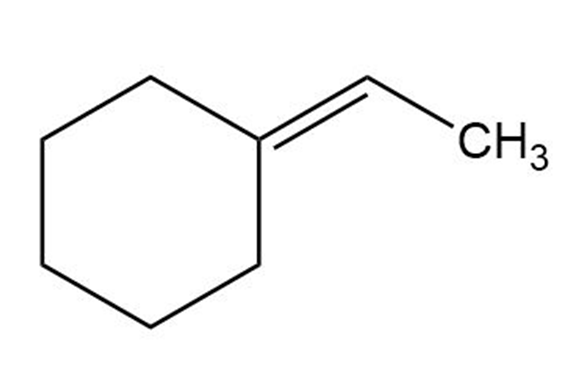
(b)
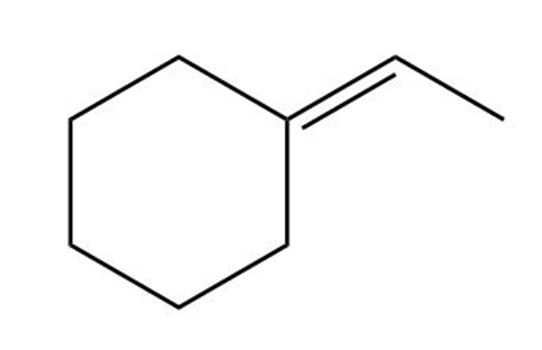
(c)
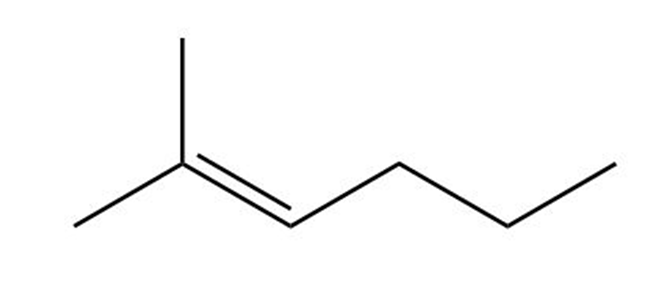
(d)
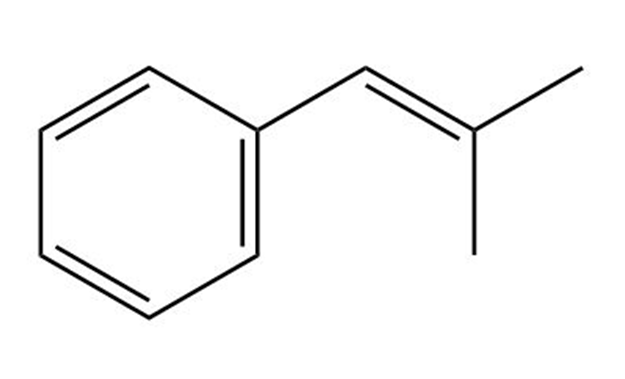
(e)

(f)

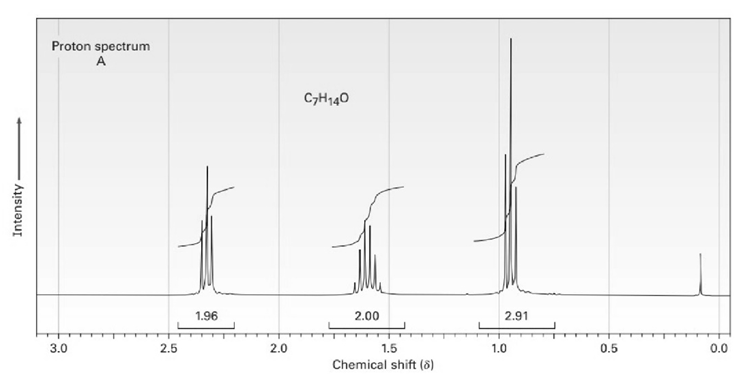
The proton and carbon NMR spectra for each of three isomeric ketones with the formula C7H14Oare shown below. Assign a structure to each pair of spectra.


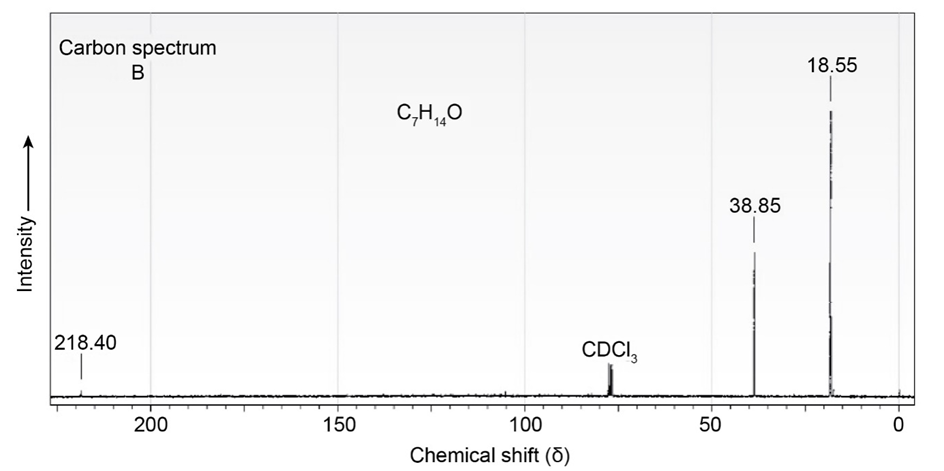
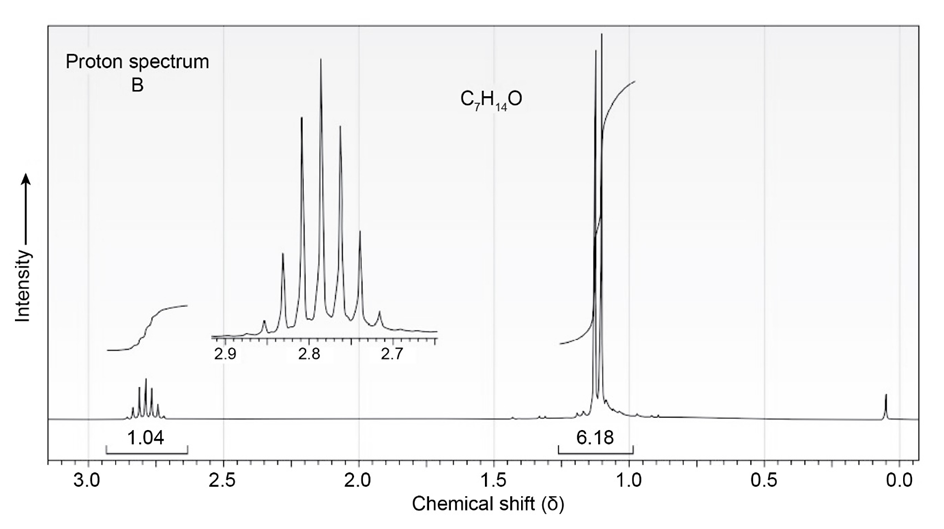
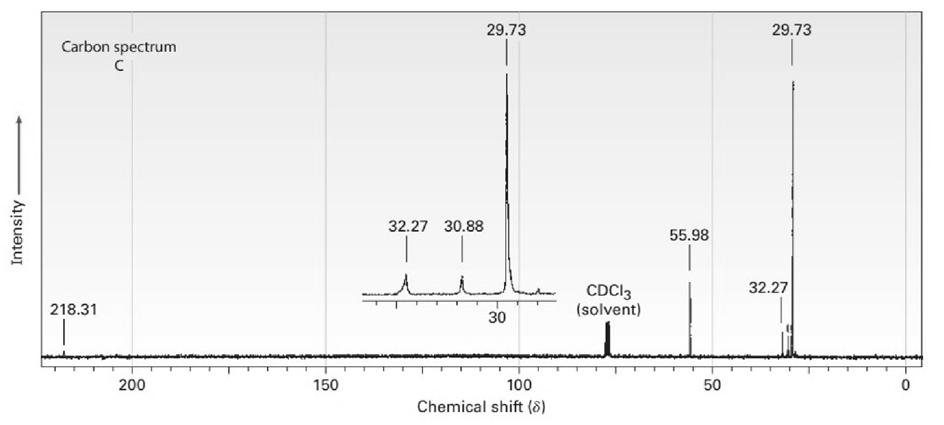
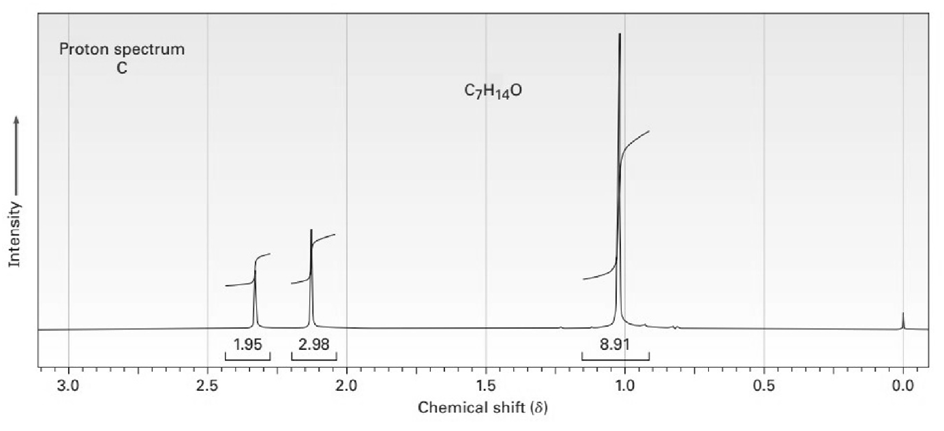
Treatment of 2-cyclohexenone with HCN/KCN yields a saturated keto nitrile rather than an unsaturated cyanohydrin. Show the structure of the product,
and propose a mechanism for the reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.